
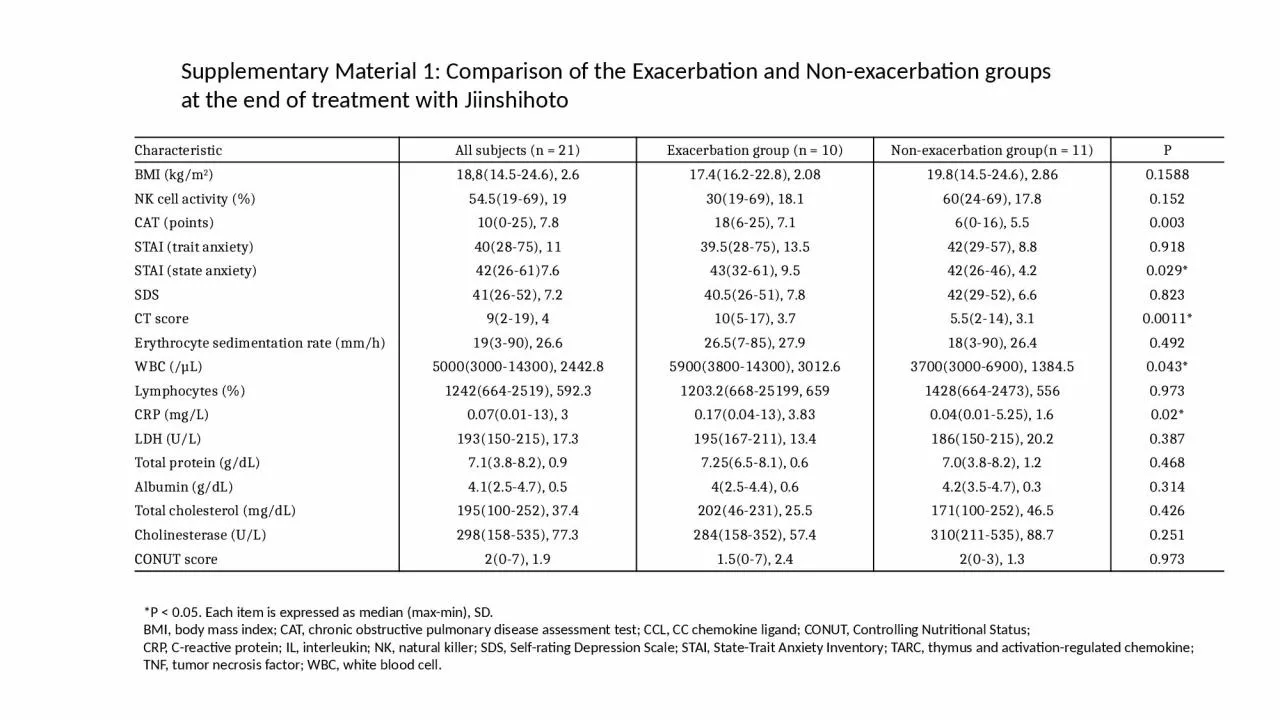
Comparison of the Exacerbation and Nonexacerbation groups at the end of treatment with Jiinshihoto Characteristic All subjects n 21 Exacerbation group n 10 Nonexacerbation groupn 11 ID: 932695
Download Presentation The PPT/PDF document "Supplementary Material 1:" is the property of its rightful owner. Permission is granted to download and print the materials on this web site for personal, non-commercial use only, and to display it on your personal computer provided you do not modify the materials and that you retain all copyright notices contained in the materials. By downloading content from our website, you accept the terms of this agreement.
Slide1
Supplementary Material 1: Comparison of the Exacerbation and Non-exacerbation groups at the end of treatment with Jiinshihoto
CharacteristicAll subjects (n = 21)Exacerbation group (n = 10)Non-exacerbation group(n = 11)PBMI (kg/m2) 18,8(14.5-24.6), 2.617.4(16.2-22.8), 2.0819.8(14.5-24.6), 2.860.1588NK cell activity (%)54.5(19-69), 1930(19-69), 18.160(24-69), 17.80.152CAT (points) 10(0-25), 7.818(6-25), 7.16(0-16), 5.50.003STAI (trait anxiety)40(28-75), 1139.5(28-75), 13.542(29-57), 8.80.918STAI (state anxiety)42(26-61)7.643(32-61), 9.542(26-46), 4.20.029*SDS 41(26-52), 7.240.5(26-51), 7.842(29-52), 6.60.823CT score9(2-19), 410(5-17), 3.75.5(2-14), 3.10.0011*Erythrocyte sedimentation rate (mm/h)19(3-90), 26.626.5(7-85), 27.918(3-90), 26.40.492WBC (/µL)5000(3000-14300), 2442.85900(3800-14300), 3012.63700(3000-6900), 1384.50.043*Lymphocytes (%)1242(664-2519), 592.31203.2(668-25199, 6591428(664-2473), 5560.973CRP (mg/L)0.07(0.01-13), 30.17(0.04-13), 3.830.04(0.01-5.25), 1.60.02*LDH (U/L)193(150-215), 17.3195(167-211), 13.4186(150-215), 20.20.387Total protein (g/dL)7.1(3.8-8.2), 0.97.25(6.5-8.1), 0.67.0(3.8-8.2), 1.20.468Albumin (g/dL)4.1(2.5-4.7), 0.54(2.5-4.4), 0.64.2(3.5-4.7), 0.30.314Total cholesterol (mg/dL)195(100-252), 37.4202(46-231), 25.5171(100-252), 46.50.426Cholinesterase (U/L)298(158-535), 77.3284(158-352), 57.4310(211-535), 88.70.251CONUT score 2(0-7), 1.91.5(0-7), 2.42(0-3), 1.30.973
*P < 0.05. Each item is expressed as median (max-min), SD.
BMI, body mass index; CAT, chronic obstructive pulmonary disease assessment test; CCL, CC chemokine ligand; CONUT, Controlling Nutritional Status;
CRP, C-reactive protein; IL, interleukin; NK, natural killer; SDS, Self-rating Depression Scale; STAI, State-Trait Anxiety Inventory; TARC, thymus and activation-regulated chemokine;
TNF, tumor necrosis factor; WBC, white blood cell.
Slide2Supplementary Material 2: Cytokine and chemokine Changes before and after the administration of
Jiinshihoto Exacerbation group (n = 10)Non-exacerbation group (n = 11)PNK cell activity (%)Before39.5(22-65, 13.945(19-67), 15.80.4594End30(19-69), 18.160(24-69), 17.80.152CCL7/TARC (pg/mL)Before578.98(21.55-1338.06), 333.79703.69(277.27-1453.13), 362.070.282End396.41(396.41-2095.76), 546.82573.53(299.94-1422.75), 327.170.259IL-2 (pg/mL)Before1.05(0.46-4.04), 1.231.05(0.46-2.23), 0.680.863End1.05(1.05-2.23), 0.851.55(1.05-3.43), 1.060.426IL-18 (pg/mL)Before162.71(73.74-488.22), 138.23137.95(54.99-220.8), 46.930.251End173.99(89.96-767.83), 255.54128.87(54.99-196.34), 41.220.051TNF-α (pg/mL)Before2.25(0.19-6.18), 1.91.89(0.7-2.95), 0.960.705End2.6(1.52-10.41), 3.082.25(0.7-4.28), 1.230.251*P < 0.05. Each item is expressed as median (max-min), SD.