
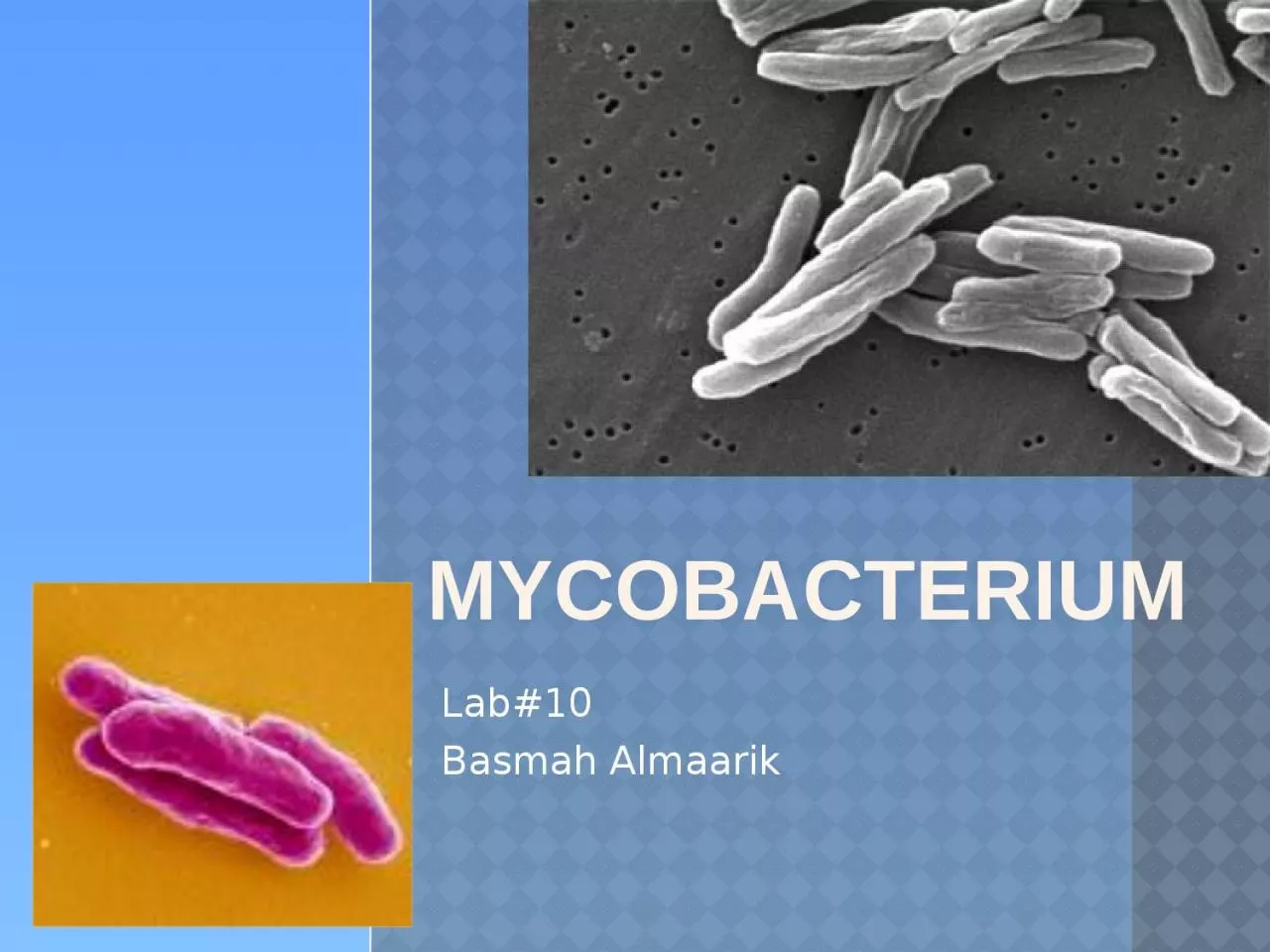
Almaarik MYCOBACTERIUM It has many species one of them is Mycobacterium tuberculosis that cause TB Some other common species seen include M avium AIDS patients M kansasii M ID: 921210
Download Presentation The PPT/PDF document "MYCOBACTERIUM Lab#10 Basmah" is the property of its rightful owner. Permission is granted to download and print the materials on this web site for personal, non-commercial use only, and to display it on your personal computer provided you do not modify the materials and that you retain all copyright notices contained in the materials. By downloading content from our website, you accept the terms of this agreement.
Slide1
MYCOBACTERIUM
Lab#10Basmah Almaarik
Slide2MYCOBACTERIUM
It has many species one of them is Mycobacterium tuberculosis that cause TB.Some other common species seen include: M. avium
AIDS patients. M. kansasii
M.
fortuitum
Slide3MYCOBACTERIUM
Specimen seen in lab are mainly sputum.Bone biopsy, CSF, urine and other.
Slide4Mycobacterium Morphology
Non spore forming, non capsulated bacilli.It has a fatty cell wall so it dose not stain well with gram stain. The gram stain can not penetrate this layer.
Acid Fast Bacilli cause it can not be decolorized with acid.
Slide5Mycobacterium Staining
Ziehl-Neelsen stain.
Kinyoun stain (cold method)
Fluorochrome
stain.
Slide6Mycobacterium Staining
Ziehl-Neelsen technique:CarbolfuchsinAcid alcohol 3%
Methylene blue
Once
Carbolfuchsin
go inside the bacterial cell it can not be decolorized by acid because of the fatty cell wall
Slide7Ziehl-Neelsen Technique
Do a regular smear.Heat fix slide.
Cover slide with carbol
fuchsin
allow heated
Stain to stay for 5 min
Need longer fixation time because of the thick fatty cell wall
a)
We
apply heat under slide until
vapor
just begins
to rise (this will allow penetration of
stain inside the cells).
b)
Or we better use heated
carbolfuschin
and if it dray we apply more of the stain
(this to avoid fire hazard)
Slide8Ziehl-Neelsen Technique
Wash with water.Now the
carbolfoschin and the fatty layer of cell wall make a complex that is red and cannot be decolorized
Cover slide with acid alcohol 3-5 mint tell all the red color is gone.
(take care cause acid alcohol is flammable)
Wash slide well with water.
Cover slide with
methylene
blue for 3-5 mint
Wash and
examin
.
Slide9Ziehl-Neelsen Technique
1
2
3
4
5
6
Slide10Ziehl-Neelsen Technique
7
Red bacilli against a blue background
Results
Very slender bacilli, arranged in single, or in pairs or in long parallel bundles making cord formation
Slide11Modifified Ziehl-Neelsen
Technique
Acid Fast Bacilli
Non- Acid Fast Bacilli
Carbolfuschin
Decolorizer
Methylin
Blue
Slide12Kinyoun Stain (Cold Method)
Same steps as Zhiehl-Neelsen but with few changes:No heating Concentrated carbolfuschin
.Concentrated decolorizing agent.Same results (red bacilli- blue back ground)
Slide13Fluorochrome Stain
When exposed to UV light it will fluorescent.These slides are examined using fluorescent microscope. If Bacilli are present it will fluorescence against a dark background.This method is used for screening if positive it is confirmed by
Common fluorochrome
stain:
Auramine-Rhodamin
stain
Acridine
orange stain
Slide14Fluorochrome Stain
1. Heat fix slide
2. Flood with
auramine
-phenol stain 10 m
3. Wash with water
4. Decolorize 1% acid alcohol 5 m
5. Wash with water
6. Cover with potassium permanganate 10 sec
Slide15Fluorochrome Stain
7. Rinse thoroughly with water
Examine at x25 or X40
Bacilli will appear yellow orange against a black background
.
Always include a positive control smear with each batch to make sure stain is ok
Slide16Fluorochrome Stain
Acridine orange stain.This fluorochrome stain can also be used for screening slide for AFB.
Staining solutions:Acridine orange stain.Alcohol saline solution.
Sodium chloride 8.5% (saline)
Slide17Examining the Slide
Fluorochrome
Stine are examined at X20 or X40 objective
Ziehl-Neelsen
and
Kinoyoun
are examined in oil immersion X100 objective
Slide18Mycobacterium Culture
The media used is Lowenstein Jensen (LJ)It is enriched mediumMycobacterium grow aerobically.35-37 ˚C.
Slow grower.M.tuberculosis raised , dry cream (buff) colonies.
Visible colonies appear after 2-3 weeks of incubation.
But culture should be incubated up to 6 weeks before discarding.
Slide19Lowenstein Jensen
Media contain:Glycerol (enhances the growth of Mycobacterium tuberculosis)
Antibiotic (Low levels of penicillin and
nalidixic
acid are also present in
LJ
medium to inhibit growth of gram positive and gram negative bacteria
)
Malachite
green
(
inhibits most other
bacteria)
Egg
yolk
Slide20Lowenstein Jensen
Coli flower appearance colonies, easily removed but very difficult to emulsify, non pigmented
Slide21Rapid methods for DetectionBACTEC
™ 12B System
Fluid medium containing
palmotic
acid carbon 14 labelled (
C14
)