
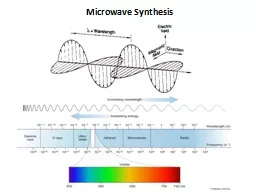
In the electromagnetic spectrum the microwave radiation region is located between infrared radiation and radiowaves Telecommunication and microwave radar equipment occupy many of the band frequencies in this region In order to avoid interference with these systems ID: 784239
Download The PPT/PDF document "Microwave Synthesis Introduction" is the property of its rightful owner. Permission is granted to download and print the materials on this web site for personal, non-commercial use only, and to display it on your personal computer provided you do not modify the materials and that you retain all copyright notices contained in the materials. By downloading content from our website, you accept the terms of this agreement.
Slide1
Microwave Synthesis
Slide2Introduction
In the electromagnetic spectrum the microwave radiation region is located between infrared radiation
and
radio-waves.
Telecommunication and microwave radar equipment occupy many of the band frequencies in this region. In order to avoid interference with these systems,the household and industrial microwave ovens operate at a fixed frequency of 2.45 GHz.[17–19]The energy of the quantum involved can be calculated by the Planck’s law E = h ν and is found to be 0.3 cal mol–1.
Slide3Conventional heating
In this method of heating, reactants are slowly activated by
a conventional
external heat source.
Heat is driven into the substance, passing first through the walls of the vessel in order to reach the solvent and the reactants. This is a slow and inefficient method for transferring energy into the reacting system.
Slide4Microwave heating
Here, microwaves couple directly with the molecules of the entire
reaction mixture
, leading to a rapid rise in the temperature.
As process is not dependant on thermal conductivity of the vessel, the result is an instantaneous localized superheating of any substancethat will respond to either dipole rotation or ionic conductivity. Only the reaction vessel
contents are
heated and not the vessel
itself
Results
better homogeneity and selective heating of polar molecules
Slide5Principles of Microwave Activation
The acceleration of chemical reactions by microwave exposure results from the interactions between
the material and electromagnetic field leading to the thermal and specific (
non-thermal) effects.
(1) Dipole interactions For microwave heating, the substance must possess a dipole moment. A dipole
is sensitive
to external electric field and tries to align itself with the field by rotation.
If submitted to
an alternating current, the electric field is inversed at each
alterance
and
therefore
dipoles
tend to
move together to follow the inversed electric field.
Such
a characteristic induces rotation
and friction
of the molecules, which dissipates as internal homogeneous heating.
Slide6The electric field of commonly used irradiation frequency (2450 MHz) oscillates 4.9 × 109 times per second.
Thus, microwave heating is directly dependent on dielectric properties of a substance, dielectric constant (ε’) and dielectric loss (ε”).
The ability of a material to convert electromagnetic energy into heat energy at a given frequency and temperature, is calculated using
ε’’ / ε’ = tan δ (1)where δ is the dissipation factor of the sample ε” is the dielectric
loss - which
measures
the efficiency
with which heat is generated from the electromagnetic radiation
and
ε’ is the
dielectric constant
which gives the ability of a molecule to be polarized by an electric field
.
The high
value of
dissipation factor δ indicates large susceptibility to microwave energy
Slide7(2) Ionic conduction
The conduction mechanism is due to the much stronger interaction of ions with electric field to generate
heat.
The ions
move under the influence of an electric field, resulting in an increased collision rate, converting kinetic energy into heat. The heat generated by both mechanisms adds up resulting in a higher final temperature and
increased reaction rates
Slide8MICROWAVE INDUCED SUPERHEATED BOILING OF SOLVENTS
Conventional heating
In organic synthesis, it is common practise to carry out reactions under reflux conditions.
The
boiling ensures a good mixing and the highest possible temperature for the solvent at atmospheric pressure. The temperature of a boiling solvent is normally assumed to be exactly at the point were the partial vapour pressure of the solvent is equal to 1 bar. However, this is not necessarily the case. During
reflux, solvent continuously evaporates, condenses and flows back into the reaction pool
.
Hence, the system is in a steady state rather than in equilibrium and the temperature is not exactly at the equilibrium boiling point.
MICROWAVE ASSISTED HETEROGENEOUS AND HOMOGENEOUS REACTIONS
Farid
CHEMAT
(1)
and Erik ESVELD
(2
)
Fifth International Electronic Conference on Synthetic Organic Chemistry (ECSOC-5), http://www.mdpi.org/ecsoc-5.htm, 1-30 September 2001
Slide9SUPERHEATING or SUPERBOILING
In a microwave heating, the average temperature of the solvent can be at
higher temperature than the atmospheric boiling point
. This is because microwave power is dissipated over the whole volume of the solvent, where nucleation points neccessary for boiling are absent.
The loss of the excess of thermal energy by boiling can therefore only occur at the side of the reactor or at the solvent-air interface
.
This
results in a reversed temperature profile with a steady average reflux temperature above the classical boiling point
.
This is called super boiling.
With
the same reactor overheating is not observed under conventional heating (
Mingos
and
Baghurst
, 1992).
Slide10Superheating advantage
The microwave superheating phenomena can be used
to accelerate homogeneous chemical
reactions
Therefore, reaction times can be reduced from days under classical heating to minutes under microwave heatingIn comparison with the classical process, the volume of the reactor, time, waste reject and the amount of solvent required are reduced.
Factors affecting microwave
heated super boiling
the physical properties of the
solvent
the
reactor
geometry
the mass
flow
the heat flow and the electric field distribution.
Among
the solvent properties, the
assosiative
properties and dielectric properties are of prime importance.
Slide11Effects of solvents in microwave
assisted synthesiscompounds
with high dielectric constants such as water,
ethanol,
acetonitrile, N,N-dimethylformamide (DMF), acetic acid, chloroform. dichloromethane, acetone, ethylene glycol etc., tend to heat rapidly under microwave irradiation.while less
polar substances
, such as aromatic and aliphatic hydrocarbons or compounds with no net
dipole moment
, such as carbon dioxide, carbon tetrachloride, diethyl ether etc.
are
poorly absorbing.
Slide12Microwave technology in process optimization
Slide13Microwave technology
applications in various organic reactions and heterocyclessynthesis
Chemists have successfully conducted a large range of organic reactions. These include :
Microwave-assisted organic synthesis is being widely applied for developing compounds in the lead optimization phase in the pharmaceuticals industry.
8.
Esterification
9.
Cycloaddition
reaction
10.
Epoxidation
11. Reductions
12. Condensations
13. Protection and
deprotection
14.
Cyclisation
reactions.
1. Diels-Alder reaction
2. Heck reaction
3. Suzuki reaction
4.
Mannich
reaction
5. Hydrogenation of [beta]-
lactams
6. Hydrolysis
7. Dehydration
Slide14Organic synthesis at atmospheric pressure
Microwave-assisted organic reactions can be conveniently conducted at atmospheric pressure in reflux conditions
e.g
. Diels-Alder reaction of
maleic anhydride with anthracene. In the presence of diglyme (boiling point 162 ºC)
this reaction can be completed in a minute, with a 90% yield
However, the conventional synthetic route, which uses benzene, requires 90 minutes.
High boiling solvents are preferred in microwave assisted organic synthetic reactions.
Slide15Organic synthesis at elevated pressure
Microwaves can be used to directly heat the reaction mixture in sealed microwave-transparent containers.
The sealed container helps in increasing the pressure in the reactor, which facilitates the reaction
This results in a substantial increase in the reaction rate
However, increase in the reaction rate of any chemical synthesis depends on three factors Volume of the vessel, the solvent to space ratio, and the solvent boiling point.
Slide16Organic synthesis in dry media
Microwaves have been applied to organic synthesis in dry media, using solid supports
i.e. alumina,
montmorillonite
clay, alkali metal fluoride doped alumina and silicaOr strongly absorbing (i.e., graphite) inorganic supportMicrowave radiation, based on solid supports, has been highly successful in reducing the reaction time E.g. condensation, acetylation and deacetylation reactions
deacetylation
of a protected compound such as alcoholic acetate held on a support material.
The microwave assisted reaction could be completed within two to three minutes, compared to conventional oil-bath heating at 75 °C for 40 hours
Slide17The first reports
Use of microwave heating to accelerate organic chemical transformations (MAOS) were published by the groups of Richard
Gedye
and Raymond J.
Giguere/George Majetich in 1986.
Slide18Applications of microwaves in heterocyclic ring formation
Five-membered
heterocyclic rings
Pyrroles:The classical Paal-Knorr cyclization of 1,4-diketones to give pyrroles is dramatically speeded- up under microwave irradiation and high yields are obtained
Slide19Imidazoles
:An important classical preparation of imidazoles is from an α-diketone, an
aldehyde
and ammonia. Here again, excellent yields can be obtained in reaction times of a few minutes as
Slide20Oxazolines
Preparation of oxazolines shows that partially saturated five-membered rings can also be prepared using microwaves
Slide21Tetrazoles
Slide22Fused heterocycles
Indoles
The classical Fischer-
indole
synthesis from an aryl hydrazine and a ketone is speededup by several 100-fold
Slide23Six-membered rings
Dihydropyridines
Slide24DihydropyrimidinesThe Biginelli reaction is important for the preparation of dihydropyrimidine
derivatives and excellent results are found for reactions carried out with microwave enhancement
Slide25Polycyclic six-membered rings
Quinolines
The
Skraup synthesis has a bad reputation as it involves very messy conditions and gives only low yields of quinolines when carried out conventionally. Recently, it has been reported that microwave enhancement reduces the reaction time to a few minutes and allows high yields to be isolated