
They can be joined together by chemical bonds From last lesson Atoms can have different numbers of protons neutrons and electrons There are 118 different types of atoms They can be found on the periodic table of elements ID: 1030580
Download Presentation The PPT/PDF document "Elements and Compounds Atoms are the fun..." is the property of its rightful owner. Permission is granted to download and print the materials on this web site for personal, non-commercial use only, and to display it on your personal computer provided you do not modify the materials and that you retain all copyright notices contained in the materials. By downloading content from our website, you accept the terms of this agreement.
1. Elements and Compounds
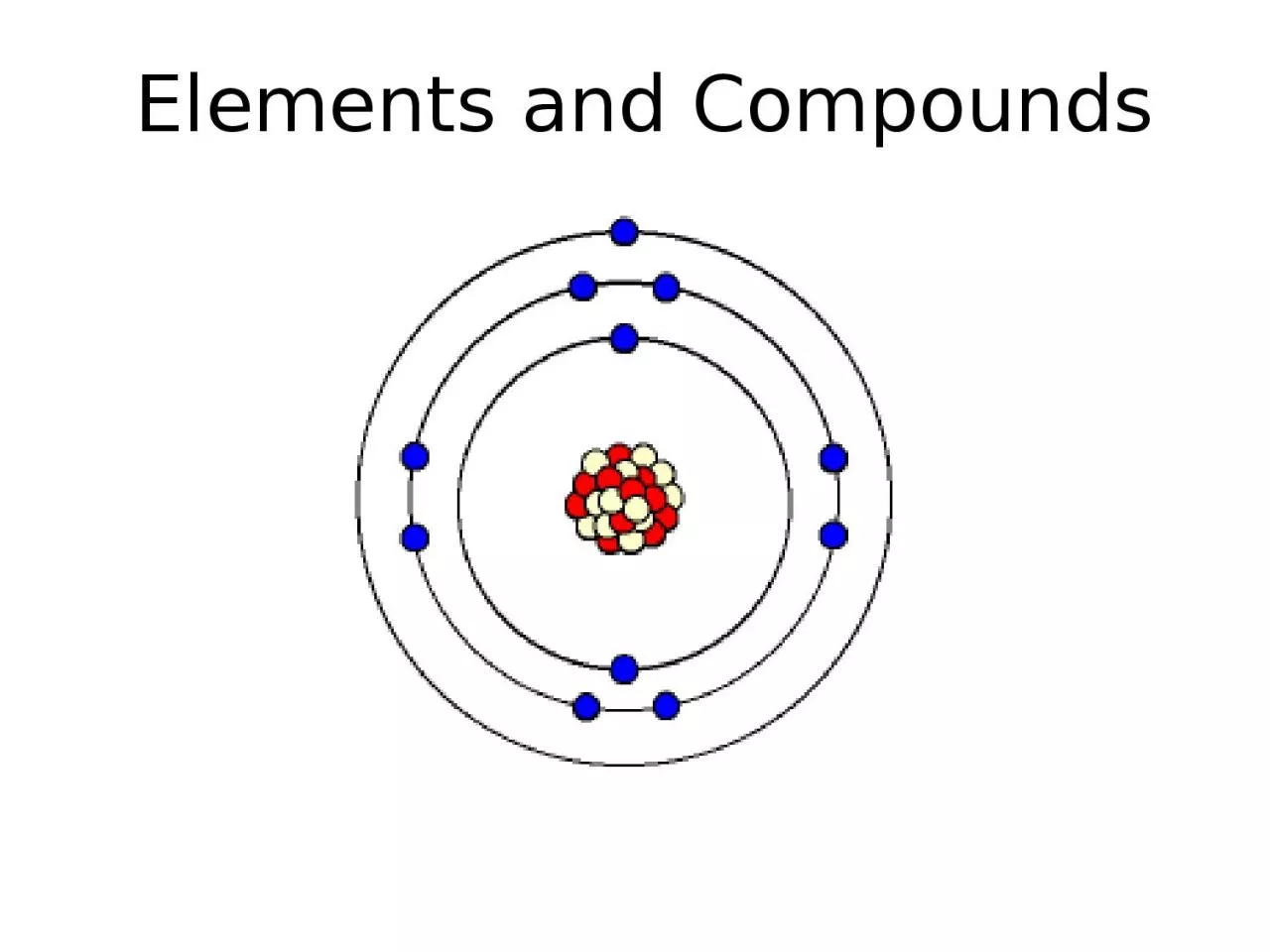
2. Atoms are the fundamental building blocks of the universe.They can be joined together by chemical bonds
3. From last lesson:Atoms can have different numbers of protons, neutrons and electrons.There are 118 different types of atoms.They can be found on the periodic table of elements.
4. If something is made of the same type of atom then it is called an element
5. Complete questions 1-9 on your sheetExtension: find the periodic table of elements in the back of your link book. Start thinking about all the things on it that you notice
6. The 118 different types of atom all have a name and a special symbolThese can be found on the Periodic Table of Elements
7. When different things are together but not chemically bonded, the result is called a mixture
8. Types of Substance
9. If between 2 and 100 atoms are chemically bonded together, the substance is called a moleculeThey can be the same atoms or different ones!
10. Q10-15 in booklet
11. Molecular Substances
12. Each substance has a chemical formula.This tells you which atoms the molecules are made ofFor example, water’s chemical formula is H2O. This means each molecule has two hydrogen atoms and one oxygen atom chemically bonded together.Molecules have between two and a hundred atoms
13. Fill in the gaps on the worksheetStart reading ahead in the worksheet
14. Atoms of which elements, and how many of them, are in:H2O2H2OCH4CO2Q17-25
15. Giant structures
16. In one kg of iron there are 1.1x1025 atomsThat means in one kg of iron there are:11000000000000000000000000 atoms
17. These atoms are all chemically bonded – billions and billions of them on and on and on. This is called a giant structure
18. Giant structures can be elements or compoundsIronIron oxide
19. Complete the next fill in the gaps on the sheetStart reading ahead
20. We can also write chemical formulae for giant structuresE.g. aluminium oxide is Al2O3 It has two aluminium atoms and three oxygen atomsQ26-33