
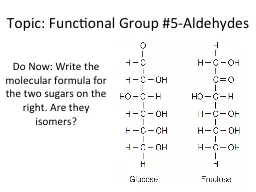
Do Now Write the molecular formula for the two sugars on the right Are they isomers General formula R C H or RCHO Carbonyl group just like ketone except always at end of aldehyde ID: 780717
Download The PPT/PDF document "Topic: Functional Group #5-Aldehydes" is the property of its rightful owner. Permission is granted to download and print the materials on this web site for personal, non-commercial use only, and to display it on your personal computer provided you do not modify the materials and that you retain all copyright notices contained in the materials. By downloading content from our website, you accept the terms of this agreement.
Slide1
Topic: Functional Group #5-Aldehydes
Do Now: Write the molecular formula for the two sugars on the right. Are they isomers?
Slide2General formula: R
C
H or RCHOCarbonyl group just like ketone except always at end of aldehyde Attached to Carbon chain on one side and a H on the other
O
Slide3Properties = same as ketones
C=O
R
H
C=O
R
H
+
-
-
+
Aldehydes are polar!
Dissolve in water
Dipole-Dipole interactions
Boiling point:
higher than alkanes (same # C
’
s)
lower than alcohols (same # C
’
s)
H H
O
+
-
Slide4Acetaldehyde (ethanal) = important aldehyde
Occurs naturally in
Coffee
BreadRipe fruitProduced by plantsProduced by the partial oxidation of ethanol in the liver and may be a contributing factor to hangovers from alcohol consumptionIt’s an air pollutant resulting from combustion (car exhaust and cigarette smoke)
Slide5Flavoring agents
Slide6Naming
Find name of alkane with same # of C
’
schange the -e to -alNever need #’s for aldehydes b/c functional group always on end C
Slide7H
C
H
O
Methanal
H
H
C
C
H
H
O
Ethanal
common name = acetaldehyde
common name = formaldehyde
Slide8CH
3
CH
2CH2CHO
CHO ending indicates
aldehyde
4 Carbons so base name is butane
Drop -e and add al
butanal
Slide9