
Antioxidant assays Total phenol content mg of GAEg of extract Total flavonoid content mg of QEg of extract DPPH radical scavenging activity ABTS radical scavenging activity Ferric reducing power assay ID: 918467
Download Presentation The PPT/PDF document "Endophytic fungus Biochemical analysis" is the property of its rightful owner. Permission is granted to download and print the materials on this web site for personal, non-commercial use only, and to display it on your personal computer provided you do not modify the materials and that you retain all copyright notices contained in the materials. By downloading content from our website, you accept the terms of this agreement.
Slide1
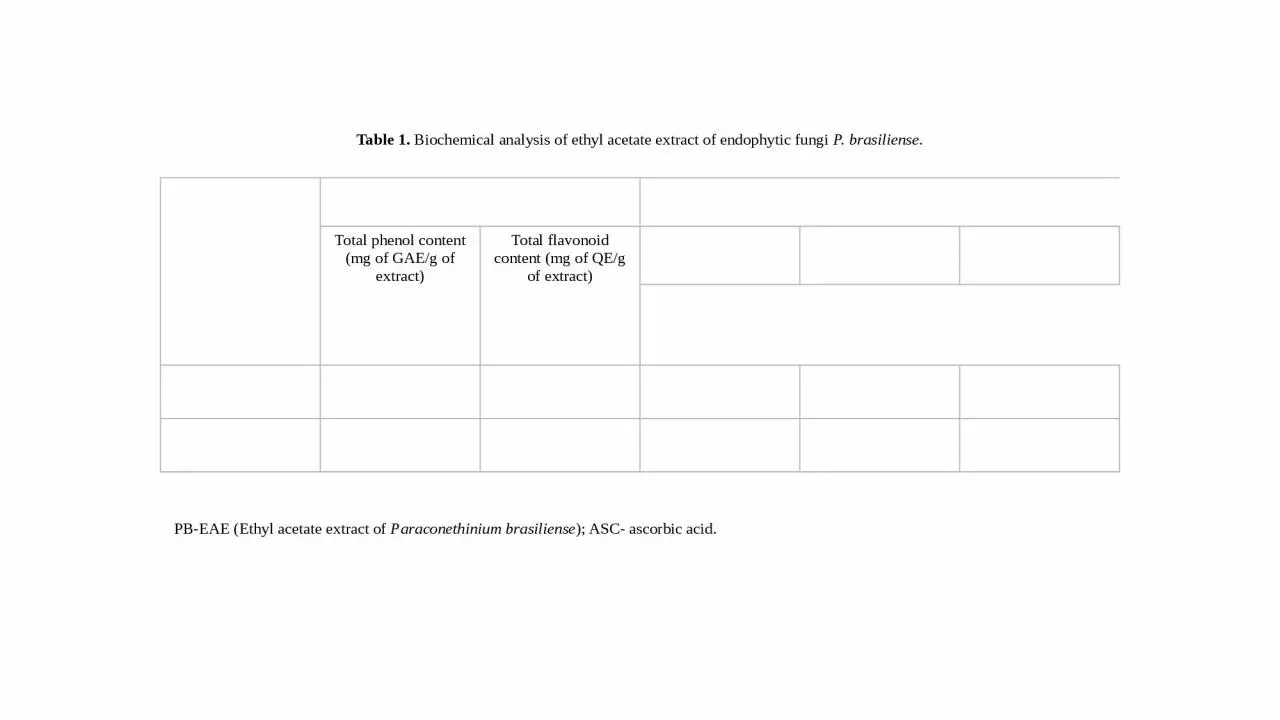
Endophytic fungus Biochemical analysisAntioxidant assaysTotal phenol content (mg of GAE/g of extract)Total flavonoid content (mg of QE/g of extract)DPPH radical scavenging activityABTS radical scavenging activity Ferric reducing power assayIC50 concentration (µg/mL)PB-EAE2.59 ± 0.06 31.53 ± 0.90383.51± 6.9729.57 ± 1.56358.17 ± 10.33ASC--69.11 ± 0.9941.84 ± 1.37317.6 ± 4.11
Table 1. Biochemical analysis of ethyl acetate extract of endophytic fungi P. brasiliense.
PB-EAE (Ethyl acetate extract of
Paraconethinium brasiliense
); ASC- ascorbic acid.
Slide2Retention TimePeak areaCompound Name (IUPAC)Molecular FormulaMolecular weight5.51314.272,3-Butylene glycolCH3CH(OH)CH(OH)CH390.127.42020.92Isopentyl acetateCH3COOCH2CH2CH(CH3)2130.1812.9551.35
o-CymeneCH
3
C6H4CH(CH3)2134.2213.120 1.73DipenteneC10H16136.2314.3722.28γ-TerpineneC10H16136.2315.5810.453,5-Nonadien-7-yn-2-olC9H12O136.1917.4214.822-PhenylethanolC6H5CH2CH2OH122.1619.9836.534-Carvomenthenol C10H18O154.2523.3876.482-Phenethyl acetateCH3COOCH2CH2C6H5164.2032.9231.70HeneicosaneCH3(CH2)19CH3296.5734.0013.402,4-Di-tert-butylphenol[(CH3)3C]2C6H3OH206.3249.7231.962,5-Piperazinedione C4H6N2O2114.10
Table 2.
GC-MS analysis of endophytic fungal extract. The possible existence of volatile compounds and its retention time, peak area, molecular formula, and molecular weight.
Slide3SampleB. cereusS. aureus L. monocytogensE.coliS. entericaEAE-PB (µg/mL)Zone of inhibition (mm)1256 ± 0.6e15 ± 1.2c14 ± 1.0c-12 ± 0.8d2508 ± 0.8d19 ± 1.4b
18 ± 1.2b -
14
± 1.0c50013 ± 0.8c20 ± 1.2ab22 ± 1.4a6 ± 0.4c18 ± 1.6b100017 ± 1.2b22 ± 1.4a24 ± 1.4a8 ± 0.4b21 ± 1.6aTCH-100021 ± 1.2a22 ±1.2a23 ±1.4a12 ± 1.2a21 ± 1.4aMinimum inhibitory concentration (µg/mL)EAE-PB31.2515.6215.6262.515.62Table 3. Antibacterial activity of ethyl acetate extract of P. brasiliense.Values represent the mean ± SD of three replicates. Values followed by uppercase letters in a column are significant differences (P < 0.05) among the groups. EAE-Pb – Ethyl acetate extract of Paraconethinium brasiliense; TCH – Tetracycline hydrochloride.
Slide4NoCompound Molecular WeightHydrogen BondLog PMolar Refractivity
Rules satisfied
Donor
Acceptor12,3-Butylene glycol90.1222-0.1823.675/52Isopentyl acetate130.18021.6337.055/53o-Cymene134.22004.4745.994/5
4
Dipentene
136.23
0
03.2747.125/55γ-Terpinene136.23003.2747.125/563,5-Nonadien-7-yn-2-ol136.19112.1943.755/572-Phenylethanol122.16111.8737.385/584-Carvomenthenol154.25112.3048.805/592-Phenethyl acetate164.20022.2947.115/510Heneicosane296.57007.60103.064/5112,4-Di-tert-butylphenol206.32113.8767.015/5122,5-Piperazinedione114.1022-1.7433.065/5
Table 4.
Drug-Likeness and toxicity prediction of volatile compounds in endophytic fungal EA extract using Lipinski’s rule.
Slide5Compound Binding energy (kcal/mol)No. of hydrogen bondsHydrogen bonding residuesIsopentyl acetate-6.971SER274o-Cymene-9.77--Dipentene-9.44-
-
γ-
Terpinene-9.22--3,5-Nonadien-7-yn-2-ol-8.681GLN2492-Phenylethanol-8.401ALA2754-Carvomenthenol-8.291SER2742-Phenethyl acetate-8.24--Heneicosane-9.17--2,4-Di-tert-butylphenol-9.27--2,5-Piperazinedione-5.204PRO15; THR53;VAL17; GLY51Table 5. Molecular modeling results for the tested compounds and 5JQ9 during docking in the Yersinia Pestisis Dihydropteroate Synthase (DHPS).