
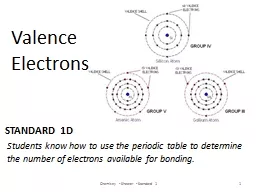
Valence Electrons Chemistry Shearer Standard 1 1 Students know how to use the periodic table to determine the number of electrons available for bonding Why are Valence Electrons Important ID: 560420
Download Presentation The PPT/PDF document "Standard 1d" is the property of its rightful owner. Permission is granted to download and print the materials on this web site for personal, non-commercial use only, and to display it on your personal computer provided you do not modify the materials and that you retain all copyright notices contained in the materials. By downloading content from our website, you accept the terms of this agreement.
Slide1
Standard 1d
Valence Electrons
Chemistry - Shearer - Standard 1
1
Students
know how to use the periodic table to determine the number of electrons available for bonding. Slide2
Why are Valence Electrons Important?Slide3
Protons and Neutrons are in the nucleus
Electrons are located in energy levels (shells) that surround the nucleus
Review…
Electrons in the outer shell of
an atom are responsible for chemical bonds!
If an outer shell holds 8 valence electrons – no bonding will take place!
Slide4
Valence Electrons
Electrons are arranged around the nucleus in 7 specific areas called energy levels.
The valence electrons participate in the formation of chemical bonds. Valence electrons are the outermost electrons.
Chemistry - Shearer - Standard 1
4
How many electrons are available for bonding?Slide5
nucleus
E
1
E
2
E
3
E
4
1
2
3
4
5
6
7
E
6
E
5
E
7Slide6
You can determine how many valence electrons
an element has by determining which group it is in.
How to Determine Valence Electrons
1
2
3
4
5
6
7
8
2Slide7
Elements with 8 valence electrons have a full outer shell and will not bond! (Noble Gases)
8 outer electrons is the ideal number of valence electrons that all atoms seem to want. All elements want to be stable like the noble gases (have 8 electrons in their outer shells).
Octet RuleSlide8
The only electrons available for bonding with other atoms are the outermost electrons.
8
Chemistry - Shearer - Standard 1
Only These
Valence Electrons and BondingSlide9
The number of outer or valence electrons can be determined by their location on the periodic table.
Chemistry - Shearer - Standard 1
9Slide10
(Group 18 or 8A) = 8 valence electrons Do not react with other atoms (inert).
Chemistry - Shearer - Standard 1
10
Noble Gases and BondingSlide11
8 e
-
Chemistry - Shearer - Standard 1
11
The Snobs of the Periodic Table
“We don’t want to bond with anyone!”Slide12
What about Helium?
Helium only has 2 electrons.
How is helium a noble gas without 8 valence electrons?
Helium only has 1 energy level.
The 1
st
energy level can only hold 2 electrons!
Thus,
helium has a full outer shell. Slide13
One valence electron available for bonding. These metals readily give this electron to another atom to form an ionic bond.
Chemistry - Shearer - Standard 1
13
Alkali Metals and BondingSlide14
Chemistry - Shearer - Standard 1
14
1 e
-
“Please take
my electron
! I really want to look like a Noble Gas!”Slide15
Two valence electrons for bonding. Gives
2 electrons away to form ionic bonds.
15
Alkaline Earth Metals and Bonding
How many electrons in the second to last shell? Slide16
2 e
-
Chemistry - Shearer - Standard 1
16
“My two electrons need to go, because my second to last shell is full!”Slide17
Most have 2
valence electrons.These metals often give these electrons to another atom to form an ionic bond.Utilize another
process to find the ve-
17
Transition Metals and BondingSlide18
2e
-
Chemistry - Shearer - Standard 1
18
“Some transition elements are
found free in nature while others give 2 electrons
!”Slide19
Group 17 or 7A = seven valence electrons. T
hese nonmetals often take one electron from another atom to form an ionic bond.
19
The Halogens and BondingSlide20
7 e
-
Chemistry - Shearer - Standard 1
20
“Gaining one electron will make my valence 8! So you better watch out cause your electron I will take!”Slide21
Also known as electron dot diagrams
Useful tool for determining how an element will bondIncludes the element symbol and a group of 1-8 dots which shows the configuration of the outer-most electron shell of the atom, also called the valence
shell.
21
Lewis DotsSlide22
Lewis Dot ExamplesSlide23