
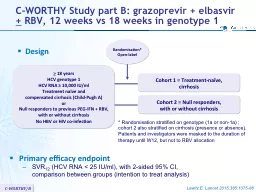
Randomisation Openlabel gt 18 years HCV genotype 1 HCV RNA 10000 IU ml Treatment naïve and compensated cirrhosis ChildPugh A or Null responders to previous PEGIFN RBV with or without cirrhosis ID: 604775
Download Presentation The PPT/PDF document "C-WORTHY/B" is the property of its rightful owner. Permission is granted to download and print the materials on this web site for personal, non-commercial use only, and to display it on your personal computer provided you do not modify the materials and that you retain all copyright notices contained in the materials. By downloading content from our website, you accept the terms of this agreement.
Slide1
C-WORTHY/B
Randomisation*
Open-label
>
18 years
HCV genotype 1HCV RNA ≥ 10,000 IU/mlTreatment naïve and compensated cirrhosis (Child-Pugh A)orNull responders to previous PEG-IFN + RBV, with or without cirrhosisNo HBV or HIV co-infection
Primary efficacy endpoint
SVR12 (HCV RNA < 25 IU/ml), with 2-sided 95% CI,comparison between groups (intention to treat analysis)
*
Randomisation stratified on genotype (1a or non-1a) ;cohort 2 also stratified on cirrhosis (presence or absence).Patients and investigators were masked to the duration of therapy until W12, but not to RBV allocation
Lawitz E. Lancet 2015;385:1075-86
C-WORTHY Study part B: grazoprevir + elbasvir + RBV, 12 weeks vs 18 weeks in genotype 1
Design
Cohort 1 = Treatment-naïve, cirrhosis
Cohort 2 = Null responders,
with or without cirrhosisSlide2
Naïve with cirrhosis
GZR + EBR + RBV = B4
GZR + EBR = B5
W18
GZR + EBR + RBV = B6
W12
GZR + EBR = B7
Cohort 1
Cohort 2
Null responders to PEG-IFN + RBV with or without cirrhosis
GZR + EBR + RBV = B8
GZR + EBR = B9
GZR + EBR + RBV = B10
GZR + EBR = B11
Treatment groups
N = 32
N = 31
N = 32
N = 31
N = 32
N = 33
N = 33
N = 29
Dosage of study drugs
Grazoprevir
(GZR) 100 mg
qd
Elbasvir
(EBR) : 50 mg
qd
RBV (bid dosing) : 800mg/day if 51-65 kg, 1000 mg/day if 66-80 kg,
1200 mg/day if 81-105 kg, 1400 mg/day if > 105 kg
C-WORTHY/B
Lawitz E. Lancet 2015;385:1075-86
C-WORTHY Study part B: grazoprevir + elbasvir
+
RBV, 12 weeks vs 18 weeks in genotype 1Slide3
GZR + EBR
+ RBV 12W
N = 31
GZR + EBR
12W
N = 29
GZR + EBR
+ RBV 18W
N = 32
GZR + EBR 18W
N = 31
Mean
age, years
57
59
59
59
Female
39%
34%
53%
32%
HCV genotype
1a
1b
other
65%
32%
3%
69%
24%
7%
75%
25%
0
74%
26%
0
HCV RNA log
10
IU/ml, mean
6.53
6.43
6.4
6.6
Discontinued treatment , N
Virologic
failure
Adverse event
3
3
0
1
1
0101110
Baseline characteristics and patient disposition(Cohort 1 : treatment-naïve with cirrhosis)
C-WORTHY/B
Lawitz E. Lancet 2015;385:1075-86
C-WORTHY Study part B: grazoprevir + elbasvir
+
RBV, 12 weeks vs 18 weeks in genotype 1Slide4
GZR + EBR
+ RBV 12W
N = 32
GZR + EBR 12W
N = 33
GZR + EBR
+ RBV 18W
N = 33
GZR + EBR 18W
N = 32
Mean
age, years
52
54
56
54
Female
37%
39%
52%
44%
HCV genotype
1a
1b
56%
44%
67%
33%
58%
42%
53%
47%
HCV RNA log
10
IU/ml, mean
6.64
6.67
6.81
6.80
Discontinued treatment , N
Virologic
failure
Adverse event
Death
2
0
1
1
3
3
0
0
1
0
001100Baseline characteristics and patient disposition(Cohort 2 : null responders with or without cirrhosis)C-WORTHY/B
Lawitz E. Lancet 2015;385:1075-86 C-WORTHY Study part B: grazoprevir + elbasvir + RBV, 12 weeks vs 18 weeks in genotype 1Slide5
SVR
12
(
HCV RNA < 25 IU
/ml) , % (95% CI)
GZR + EBR + RBV 12W GZR + EBR 12W
GZR + EBR + RBV 18W
GZR + EBR 18W
Early discontinuation
0
0
1
0
2
0
0
0
Virologic
breakthrough
1
0
0
0
0
0
0
1
Relapse
2
1
0
2
0
3
0
0
%
Cohort 2 with cirrhosis : SVR
12
= 92% with 12W and 100% with 18W ; 94% for GT1a and 100% for GT1b
C-WORTHY/B
Lawitz E. Lancet 2015;385:1075-86
C-WORTHY Study part B: grazoprevir + elbasvir
+
RBV, 12 weeks vs 18 weeks in genotype 1
0
20
40
60
80
100
Treatment-naïve (Cohort 1)
Null responders (Cohort 2)
94
(79-99)100(89-100)90 (74-88)97(82-100)97(84-100)91(76-98)N2932333297(84-100)3194(79-99)323331Slide6
GZR + EBR + RBV 12W
GZR + EBR 12W
GZR + EBR + RBV 18W
GZR + EBR 18W
Serious adverse events
2 (3%)
3 (5%)
1 (2%)
2 (3%)
Discontinuation due to AE
1 (2%)
0
1 (2%)
0
Death
1 (2%)
0
0
0
Adverse events in ≥ 10%
Fatigue
24%
23%
37%
21%
Headache
17%
18%
26%
32%
Asthenia
14%
9%
16%
14%
Hemoglobin
< 10 g/dl
10%
0
8%
0
Elevation of bilirubin > 2.5 to 5.0 x baseline
11%
0
28%
6%
Elevation of bilirubin > 5 x baseline
2 (3%)
0
1 (2%)
0
Elevation of ALT or AST > 2.5 x baseline
0
0
1 (2%)
5 (8%)
Adverse events and laboratory abnormalities, N (%)
C-WORTHY/B
Lawitz E. Lancet 2015;385:1075-86
C-WORTHY Study part B: grazoprevir + elbasvir
+
RBV, 12 weeks vs 18 weeks in genotype 1Slide7
SVR
12
according to detection of resistance-associated variants at baseline
NS3 variants at baseline
NS5A variants at baseline
NO
YES
NO
YES
N
169
79
209
34
SVR
12
(HCV RNA < 25 IU/ml)
96.4%
92.4%
97.1%*
82.4% *
Mutations detected
Most prevalent NS3 variants
at failure :
Y56H
A156T/G/V
D168A/Y
Most prevalent NS5A variants
at failure :
M28T
Q30L/R
L31M
Y93H/N
* p < 0.001
C-WORTHY/B
Lawitz E. Lancet 2015;385:1075-86
C-WORTHY Study part B: grazoprevir + elbasvir
+
RBV, 12 weeks vs 18 weeks in genotype 1Slide8
C-WORTHY Study part B: grazoprevir + elbasvir
+ RBV, 12 weeks vs 18 weeks in genotype 1
SummaryIn this phase II study, oral treatment with grazoprevir and elbasvir
, with or without RBV, in HCV genotype 1-infected patients that are difficult to cure with HCV therapy (patients with well compensated cirrhosis and null responders with or without well compensated cirrhosis), high rates of SVR
12 were shown across all groups, irrespective of the addition of RBV or extension of treatment duration from 12 to 18 weeks12 weeks of GZR + EBR without RBV achieved SVR12 of
97% in previously untreated patients with cirrhosis, 91% in null responder patients with or without cirrhosis, 92% in null responder patients with cirrhosisThe rate of virologic failure with GZR + EBR with or without RBV was low (4%)Similar efficacy was seen in patients with genotype 1a and 1bPatients with NS5A baseline resistance-associated variants had lower SVR12Treatment-emergent, clinically significant, adverse events were infrequent
No discontinuation for AE in the groups without RBV
C-WORTHY/BLawitz E. Lancet 2015;385:1075-86