
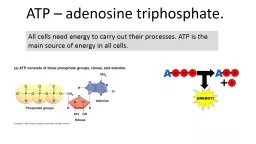
All cells need energy to carry out their processes ATP is the main source of energy in all cells What is it made of 1x adenine nitrogen containing base Ribose 5 carbon sugar 3x phosphate groups in a chain ID: 437200
Download Presentation The PPT/PDF document "ATP – adenosine triphosphate." is the property of its rightful owner. Permission is granted to download and print the materials on this web site for personal, non-commercial use only, and to display it on your personal computer provided you do not modify the materials and that you retain all copyright notices contained in the materials. By downloading content from our website, you accept the terms of this agreement.
Slide1
ATP – adenosine triphosphate.
All cells need energy to carry out their processes. ATP is the main source of energy in all cells. Slide2
What is it made of?
1x adenine (nitrogen containing base)
Ribose – 5 carbon sugar.3x phosphate groups in a chain.Slide3
Why is ATP the main source of energy?
Bonds between 3 phosphate groups are unstable and have a LOW ACTIVATION energy to break them apart.
When they break apart they release lots of energy.Slide4
The last (terminal) phosphate is usually the one to be removed.
ATP + H2O
ADP + Pi + ENERGY
The reaction.
Adenosine
tri
phosphateAdenosine diphosphateInorganic phosphate (that is lost from ATP)TIP:Tri = 3Di = 2Water is used to break apart this reaction, is it a hydrolysis or condensation reaction?Slide5
How to form ATP.
Water is released to form ATP, is it a hydrolysis or condensation reaction?
ATP + H2O ADP + Pi + ENERGYThe reaction.
Adenosine
tri
phosphate
Adenosine diphosphateInorganic phosphate (that is lost from ATP)The reaction is actually reversible. The enzyme ATP synthase is needed to make ATP.Slide6
ATP – immediate energy source.
Because it is so unstable and quick to release energy, ATP is not a good energy store.Instead it is an immediate energy source.Why is ATP an immediate energy
soure:ATP energy is released rapidlyEnergy released in a single stepTo unstable to be a store
Energy transferred directly to the reaction that needs it. Slide7
Where is ATP made and used?
ATP is made in the mitochondria of cells.
Uses:Movement - ATP provides energy for muscle contraction. (Muscle cells have loads of mitochondria.
Metabolic processes – provides energy to build big molecules.
Active transport – to move particles against a concentration gradient.
Secretion of molecules.
To activate molecules in reactions (make reactions happen).Slide8
Water and its functions. Slide9
Water = H2
OHydrogen x 2
Oxygen x 1Slide10
As a complete molecule, water has no charge, it is neutral….
But look closer and the different parts of small amounts of charge, this makes water dipolar (“two charges”)..
Oxygen = slightly negative.
hydrogen= slightly positive.
Dipolar water molecule.Slide11
The opposite charges attract and form hydrogen bonds.
This allows water to “stick together” (look at the droplets below).
Drawn like this!Slide12
Latent heat of vaporisation – energy to evaporate 1 gram of water.
All the hydrogen bonds between each water molecule means lots of energy is needed to separate them and allow them to evaporate.
To turn water to a gas all the hydrogen bonds must break first! This is why the latent heat of vaporisation is so high.
Sweat is mainly water. Sweating cools you down as loads of energy is removed from the body to make the sweat evaporate. . Slide13
Specific heat capacity of water – energy required to heat a 1kg of water by 1 degrees Celsius.
All the hydrogen bonds between each water molecule means lots of energy is needed to warm up water.
This means water stays at a relatively constant temperature, even on hot and cold days.
This protects aquatic animals from sudden changes in their environment. Slide14
Surface tension.
The hydrogen bonds connecting water means it can act like a skin.
Small insects can walk on it.Slide15
Water sticks together because of hydrogen bonds – this is called cohesion.
Cohesive forces allow water to be pulled (sucked) up a straw or the xylem of a plant.Slide16
Other uses of water.
Solvent – water dissolves other molecules.
Metabolism – chemical reactions take place in an aqueous medium (water)Metabolism – water used to break down molecules (hydrolysis).
Water is used in reactions (photosynthesis).