
Unablatable Lung Metastases A Phase I Clinical Trial Boas FE et al Published Online August 31 2021 httpsdoiorg101148radiol2021210213 In a prospective study of 10 participants all had ID: 910525
Download Presentation The PPT/PDF document "Bronchial or Pulmonary Artery Chemoembol..." is the property of its rightful owner. Permission is granted to download and print the materials on this web site for personal, non-commercial use only, and to display it on your personal computer provided you do not modify the materials and that you retain all copyright notices contained in the materials. By downloading content from our website, you accept the terms of this agreement.
Slide1
Bronchial or Pulmonary Artery Chemoembolization for Unresectable and
Unablatable
Lung Metastases: A Phase I Clinical Trial
Boas FE et al. Published Online:
August 31, 2021https://doi.org/10.1148/radiol.2021210213
In a prospective study of 10 participants, all had
hypervascular lung metastases that were supplied by the bronchial artery in nine participants (90%) and by the pulmonary artery in one participant (10%).After chemoembolization, the intratumoral mitomycin concentration was 380 times the plasma mitomycin concentration.Drug release from the ethiodized oil and mitomycin emulsion occurred with a half-life of more than 5 hours in vivo and 7.1 hours in vitro.
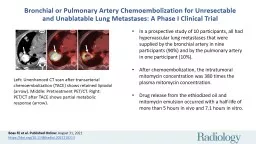
Left: Unenhanced CT scan after transarterial chemoembolization (TACE) shows retained lipiodol (arrow). Middle: Pretreatment PET/CT. Right: PET/CT after TACE shows partial metabolic response (arrow).