
Kontur W S et al A heterodimeric glutathione S transferase that stereospecifically breaks lignins βRaryl ether bond reveals the diversity of bacterial β etherases ID: 933696
Download Presentation The PPT/PDF document "Discovery of a new type of bacterial enz..." is the property of its rightful owner. Permission is granted to download and print the materials on this web site for personal, non-commercial use only, and to display it on your personal computer provided you do not modify the materials and that you retain all copyright notices contained in the materials. By downloading content from our website, you accept the terms of this agreement.
Slide1
Discovery of a new type of bacterial enzyme able to cleave bonds in lignin
Kontur
, W. S.
et al. “A heterodimeric glutathione S-transferase that stereospecifically breaks lignin’s β(R)-aryl ether bond reveals the diversity of bacterial β-etherases.” Journal of Biological Chemistry 294, 1877-1890 (2019) [DOI: 10.1074/jbc.RA118.006548].
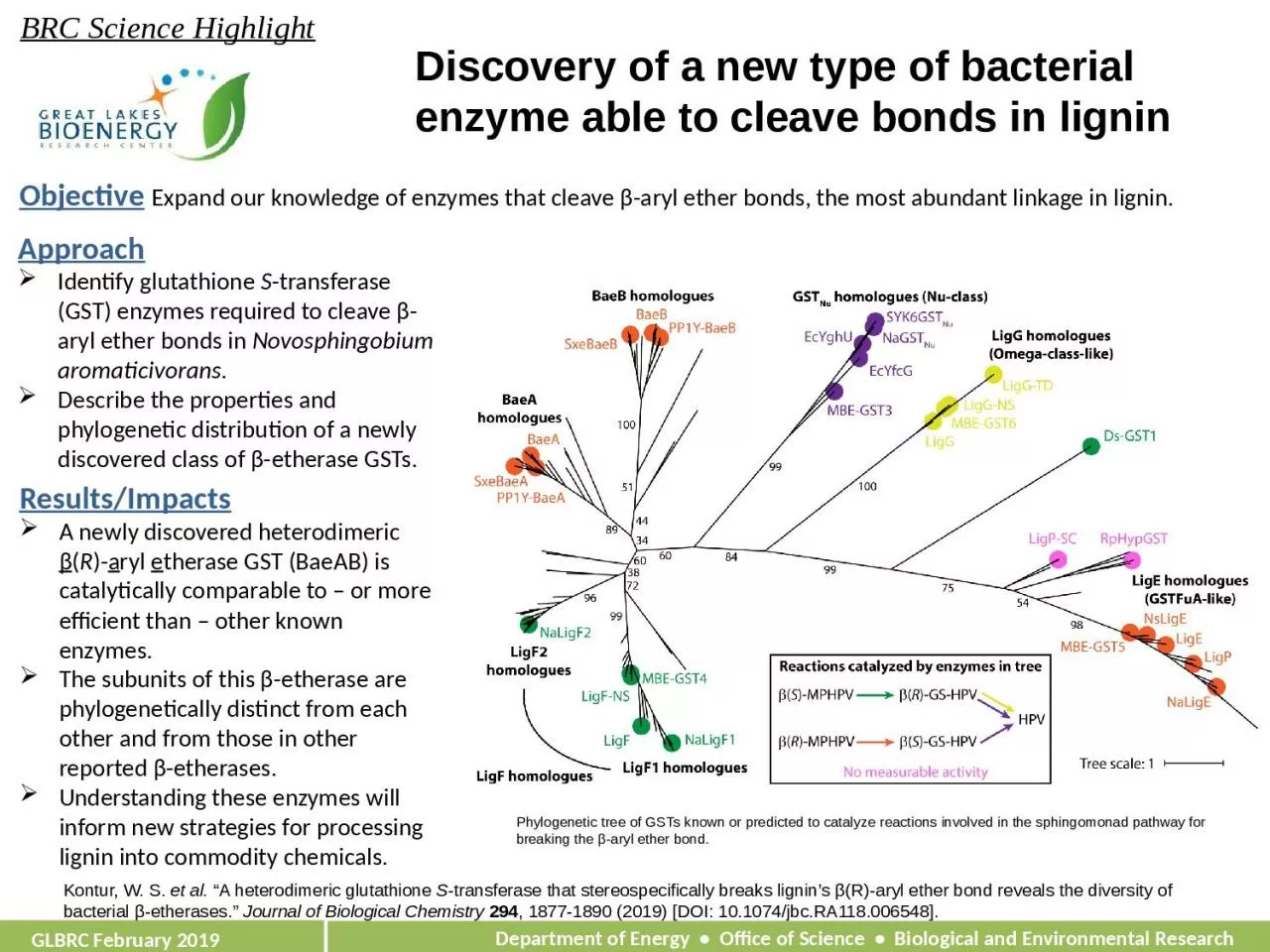
Objective Expand our knowledge of enzymes that cleave β-aryl ether bonds, the most abundant linkage in lignin.
ApproachIdentify glutathione S-transferase (GST) enzymes required to cleave β-aryl ether bonds in Novosphingobium aromaticivorans.Describe the properties and phylogenetic distribution of a newly discovered class of β-etherase GSTs.
Results/ImpactsA newly discovered heterodimeric β(R)-aryl etherase GST (BaeAB) is catalytically comparable to – or more efficient than – other known enzymes.The subunits of this β-etherase are phylogenetically distinct from each other and from those in other reported β-etherases.Understanding these enzymes will inform new strategies for processing lignin into commodity chemicals.
BRC Science Highlight
GLBRC February 2019
Phylogenetic tree of GSTs known or predicted to catalyze reactions involved in the
sphingomonad pathway for breaking the β-aryl ether bond.