
after human tumor initiation and why this might be important Darryl Shibata Department of Pathology University of Southern California Keck School of Medicine Los Angeles CA dshibatauscedu 10 ID: 946640
Download Pdf The PPT/PDF document "Is it possible to reconstruct the first ..." is the property of its rightful owner. Permission is granted to download and print the materials on this web site for personal, non-commercial use only, and to display it on your personal computer provided you do not modify the materials and that you retain all copyright notices contained in the materials. By downloading content from our website, you accept the terms of this agreement.
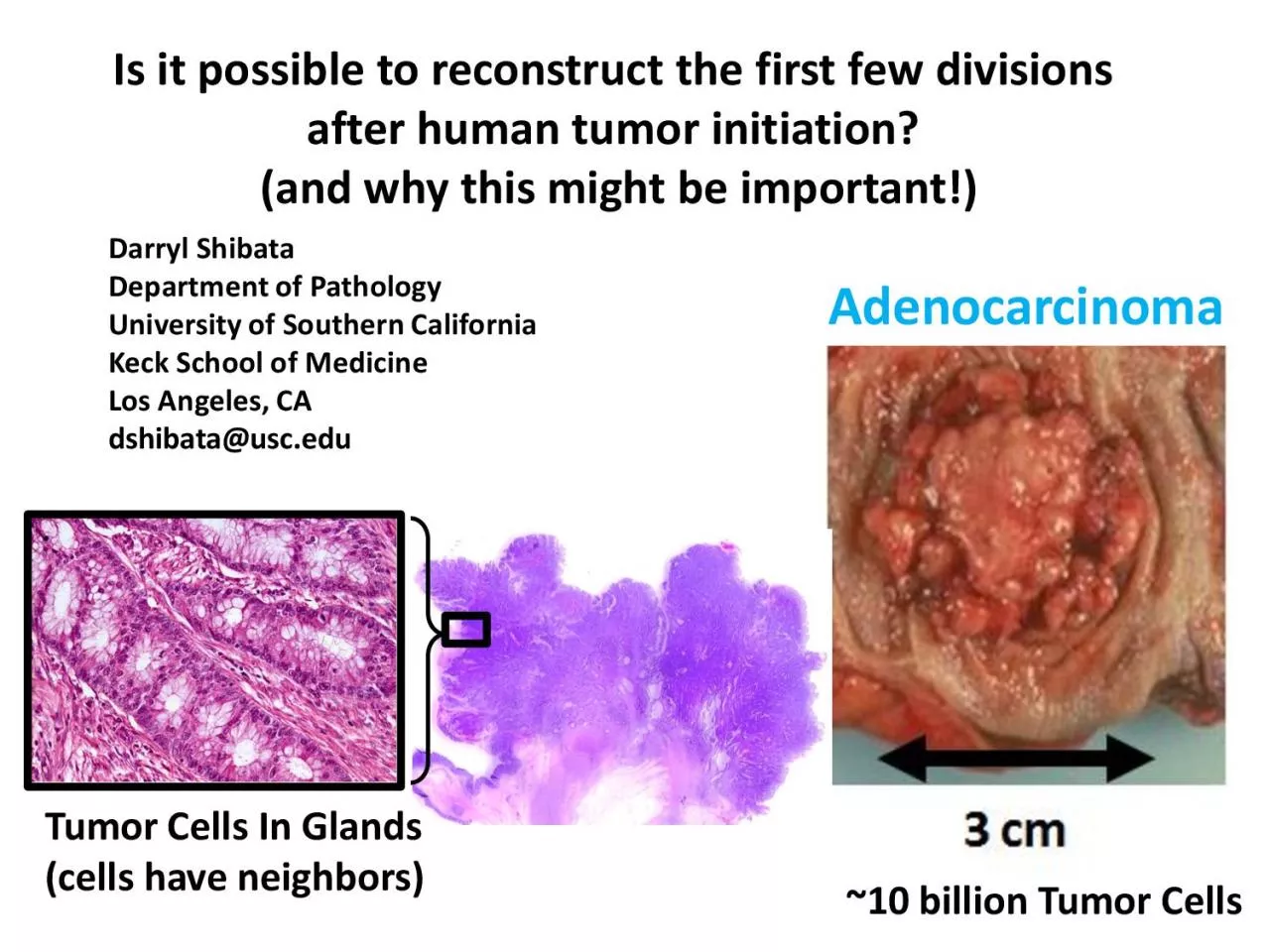
Is it possible to reconstruct the first few divisions after human tumor initiation? (and why this might be important!) Darryl Shibata Department of Pathology University of Southern California Keck School of Medicine Los Angeles, CA dshibata@usc.edu ~10 billion
Tumor Cells Tumor Cells In Glands (cells have neighbors) Adenocarcinoma What Happens During The First Few Tumor Cell Divisions? (tumors are clonal : all start from a single cell) 1. Nothing Special: Just Another Cell Division 2. Something âSpecialâ (nothi
ng normal about it) âSpecialâ UnknownsÍ --- Increased Chromosomal Instability? --- Increased Mutation Rate? --- Changes in Cell Mobility? --- Born To Be Bad? (Ability to Invade and Metastasize Already Present?) --- ????? âSpecialâ KnownÍ Initial Growth
(both daughter cells survive and divide) first tumor cell first tumor cell division Why Is It Important To Study What Happens During The First Few Tumor Cell Divisions? 1. Early Cancer Prevention: BePPer UndersPMnding Of WOMP To âPreQenPâ 2. Distinguish
Benign Versus Malignant Small Tumors (Born To Be Bad): Are The First Few Divisions Of Malignant And Benign Tumors Different? 3. Measurable (?): TOe FirsP Few Tumor Fell GiQisions Are âEMsyâ To MeMsure first tumor cell first tumor cell division For FunÍ A
âPhysicsâ Type Of âStoryâ Concept Physics Cancer BIG BANG QUANTUM MECHANICS Time (relativity) âStartâ Single Cell (13 billions yrs ago) ( clonal ) Energy In Stable Mutations In Stable Discrete Packets Discrete States (Fixation) Uniform Age U
niform Age (Speed of Light Constant) (Tumor Cell Mitotic Age Constant) Big Bang Universe Big Bang Tumorigenesis Tumors Typically Exhibit Gompertzian G rowth initial fast exponential expansion (inflation) rate slows when� 1 cm 3 1 st transformed cell Ob
servations of Tumor Growth early binary exponential division one start with billions of tips Complex Ancestral Somatic Cell Tumor Tree Complex Ancestral Somatic Cell Tumor Tree Start From Single Cell With Early Exponential Growth = Star - like Phylogeny (cells
with similar mitotic ages) Many Possible Binary Trees: BUT Early Tree Structure Relatively Is Simpler And Easy To âMeasureâ Sampling From âOppositeâ Tumor Sides Can Identify Early Private Mutations How To Measure The âStartâ Of The Universe? Big B
ang 1. First Few Seconds Of The Universe Extremely Well - characterized 2. Physicists Must Be Really Smart!!!! Primary Evidence Is The Background Temperature Or Glow of the Universe (3 degrees Kelvin) Microwave Radiation (Static) Is Uniform Or The Same In Every
Direction Uniformity Is Due To Mixing And Rapid Early Expansion (inflation) AlternativelyÍ A âStartâ Is Easy To Characterize Because The Signal Is âEverywhereâ Public Mutations (all tumor cells) The Tumor âStartâÍ Signal Is Early Private Mutations
1) Easy To Sample 2) Easy To Detect simple exponential expansion Public: 100% cells Private: Division 1: 50% Division 2: 25% Division 3: 12.5% Division 4: 6.25% Division 5: 3% NGS Platforms: Sensitivity About 10% Mutation Frequency Private Mutation (some tumo
r cells) zygote initiating cell Left Tumor side Right Tumor side private mutations private mutations public mutations public mutations private mutations Model System: Human Colorectal Cancer Specific Goal: Understand Tumor âInitiationâ (first few divisi
ons after transformation) Clinical Questions: How Do We Prevent Cancers? Are Tumors âBorn To Be Badâ? Colorectal Cancers Have Structure ( Adenocarcinomas With Glands) Tumor Gland Fragments (~10,000 adjacent cells, �95% pure) Single Tumor Gland/Frag
ment Analysis (cell neighbor analysis) 1. Chromosome Copy Number Alterations (CNA, SNP - chips) 2. DNA Passenger Methylation Patterns ( bisulfite sequencing) 3. Targeted Resequencing ( AmpliSeq / IonTorrent ) ~ 10,000 Adjacent Tumor Cells microfuge tube Relativ
e Error and Mitotic Rates (âmolecular clocksâ) DNA base fidelity ~10 - 9 per base per division DNA methylation ~10 - 5 per base per division Chromosome CNA ~10 - 2 to 10 - 4 per division Stepwise Changes Individual Glands (~10,000 adjacent cells)
Tumor Growth: 1) Cells Divide 2) But Growth is Through Gland Division (Fission) Therefore Glands May Be Stable Physical StructuresÍ Glands Can âAgeâ (their cells become polymorphic) right side 5â 3â 5â 3â 5â 3â six cancer glands 5â 3â left
side 5â 3â 5â 3â six cancer glands Experimental Strategy: Sample Multiple Tumor Glands DNA Passenger Methylation Patterns 1 st transformed cell Tumor Simulation Passenger DNA Tumor Gland Methylation: More Consistent With A Star Phylogeny (single clonal
expansion) Palm Tree - shaped âBig Bangâ (exponential clonal expansion) Star Phylogeny 1. Gland Are âOldâ or Diverse Populations (Stable) 2. Individual Glands Are Almost As Old or Diverse As Their Tumors 3. No Evidence of New or Old Parts (Equally Old o
r Young) fission Exponential Growth (32 divisions) 1 st cancer gland N o G rowth Left Side Right Side Tumor Star Phylogeny: Inherent Non - Chaotic Mechanism To Generate Variation (ITH) Growth (short time) âAgingâ (long time) Chromosome CNAs (Chromosomal In
stability (CIN)) FISH: CIN PRESENT (different ploidy ) Gland Stepwise Chromosomal Changes: Gains and Losses SNP Microarray: Average of gland = âInteger Valueâ 2N Tumor Gland âQuantum Mechanicsâ 1 st transformed cell Tumor Simulation fission Exponentia
l Growth (32 divisions) 1 st cancer gland N o G rowth 2N Why Are Tumor Glands âQuantumâ? Model: 1) Single Cell Start is Quantum (CN is integer, 0,1,2,3,4ÍÍÍ) 2) Glands Are Formed Soon After The Start And Therefore Tend To Be âQuantumâ 2 2 3 2 2 2 2
2 2 frequency 2 3 4 1 0 Cell CN 1 2 3 2 2 1 2 3 2 2 frequency 3 4 1 0 Cell CN 2 2 3 2 2 1 2 2 2 2 frequency 3 4 1 0 Cell CN Average gland CN = âstartâ Gland CN Quantum Gland CN âQuantum MechanicsâÍ Certain âStatesâ Are Favored 0 0.5 1 0 1 2 3 4 5 6
Chromosome Copy Number B - allele Frequency ( mBAF ) 1N 2N 2N LOH 3N Cells Are Inherently Quantum Glands Are Mixtures of Cells Hydrogen Atom Closely Related 0 0.5 1 0 1 2 3 4 5 6 Chromosome Copy Number Visualizing Gland Chromosome Ploidy (âquantum integer no
rmal valuesâ) B - allele Frequency ( mBAF ) SNP microarrays 1N 2N 2N LOH 3N 0 0.5 1 0 1 2 3 4 5 6 mBAF Chromosome Copy Number Despite âCINâ Most Gland Chromosome Fragments Are âFixedâ (near âquantumâ or integer values) Adenoma (8 glands) 0 0.5 1 0
1 2 3 4 5 6 mBAF Chromosome Copy Number Cancer (8 glands) 1 st transformed cell Tumor Simulation fission Exponential Growth (32 divisions) 1 st cancer gland N o G rowth Left Side Right Side 0 0.2 0.4 0.6 0.8 1 0 1 2 3 4 5 6 BAF CN Tumor Simulations of CIN Star
t: 2 chromosomes, 200 divisions Random CNA 0 0.2 0.4 0.6 0.8 1 0 1 2 3 4 5 6 BAF CN Force CNA (+1) At First Division 0 0.5 1 0 1 2 3 4 5 6 BAF Chromosome Copy Number Despite âCINâ Most Gland Chromosome Fragments Are âFixedâ (near âquantumâ or integ
er values) Adenoma (8 glands) 0 0.5 1 0 1 2 3 4 5 6 BAF Chromosome Copy Number Cancer (8 glands) Summary of Tumor Gland Alterations 1) Passenger Methylation Patterns: Diverse 2) FISH Chromosome CNAs: Diverse 3) SNP MicroarrayÍ Many Average Gland CNAs Are âQu
antumâ (reflect CN of the first tumor cells) Individual Tumor Glands: Relatively Old Stable Populations (single clonal expansion) Public Mutations (all tumor cells) What About Point Mutations? simple exponential expansion Public: 100% cells Private: Divisio
n 1: 50% Division 2: 25% Division 3: 12.5% Division 4: 6.25% Division 5: 3% Whole Tumor Single Gland Public: 100% cells Private: 0 to 100% Possible Gland Point Mutation Frequencies 1) Infinite Possible Values (0 to 100%) ---- Genomic Instability ---- Migration
and Mixing 2) âQuantumâ Values (1N, 2N, 3NÍÍÍ) ---- Detectable Mutations Are Public and Early Private Mutations ---- Individual Glands Are Old, Stable Populations (fixation or lost) (Hint: Gland Chromosome CN and Detectable Point Mutation Frequencies
Are Entangled) zygote initiating cell Left Tumor side Right Tumor side private mutations private mutations public mutations public mutations private mutations Experimental Approach 1) Bulk Sample Opposite Tumor Sides 2) NGS ( Illumina , Exome Sequence, 50X)
3) Identify Public and Private Point Mutations ( MuTec , Somatic Sniper) 4) Resequence Mutations In Bulk Sample and Individual Glands ( AmpliSeq , IonTorrent ~100X+ coverage) 0 2 4 6 8 10 12 14 16 18 20 0 0.2 0.4 0.6 0.8 1 Number of Genes Mutation Frequency B
ulk Resequencing Data: Continuous Mutation Frequencies 0 20 40 60 80 100 120 140 0 0.2 0.4 0.6 0.8 1 Number of Genes Mutation Frequency Gland Resequencing DataÍ âQuantumâ Mutation Frequencies 1/2N 1/4N 1/3N 2/3N 2/2N 0 0.2 0.4 0.6 0.8 1 0 0.2 0.4 0.6 0.8
1 Measured Mutation Frequency Expected Mutation Frequency Mutation Frequency With Respect To Ploidy 4N 3N 2N 3N 1N 0 0.2 0.4 0.6 0.8 1 0 0.2 0.4 0.6 0.8 1 Expected Mutation Frequency Bulk Tumor Individual Glands BLACK Symbols = Public Mutations RED Symbols =Pri
vate Mutations 4N 3N 2N 3N 1N Public Mutations (all tumor cells) Summary of Tumor Gland Alterations 1) Passenger Methylation Patterns: Diverse 2) FISH Chromosome CNAs: Diverse 3) SNP MicroarrayÍ Many Average Gland CNAs Are âQuantumâ 4) Mutation Resequenci
ng Í âQuantumâ or âFixedâ Detectable Point Mutation Frequencies How Did A Gland Cross To The Other Tumor Side? private mutations public Right Left Cells Migrate But Glands Donât Migrate Much How Did A Gland Cross To The Other Tumor Side? private muta
tions public Right Left A A A A A A A B B B B B B B Right How Does A Private Mutation End Up On Both Sides Of A Tumor? Left private mutations public Cells Migrate But Glands Donât Migrate Much ADENOMA ADENOMA CANCER CANCER âBorn To Be Badâ What is âBadâ
Clinically?Í Death How Do Tumors Kill? 1) Invasion 2) Metastasis Common Requirement of Invasion and Metastasis: Abnormal Cell Mobility Initiating Cell First Few Cells Do The First Few Tumor Cells Move/Mix? Late Event Novelli M et al. PNAS 2003;100:3311 - 33
14 Cell Proliferation And Movement Is Normal But Cell Intermixing Is Abnormal Development: Clonal Patches G6PDH expression: X - linked inactivation during human development âBorn To Be Goodâ Intestinal Crypts: Cell Migration in Orderly Columns âBorn To B
e Goodâ Cell Proliferation And Movement Is Normal But Cell Intermixing Is Abnormal Born To Be Good/Bad Effects of Early Cell Mixing Initial exponential expansion final benign tumor (mutation patches) left right final malignant tumor (polka dots) left right
1 st transformed cell âborn to be goodâ âborn to be badâ 1 mm movement 1 mm movement mutation patches mutation patches mutation polka dots Colorectal Adenocarcinoma -- Individual Cells Can Migrate -- Hard For Glands To Migrate Gland Scatter During Grow
th Late Private Mutation Early Private Mutation Early Private Mutation (Cell Mixing) Detectability : Human Tumors Are Large Versions Of Their Small Tumors Right How Does A Private Mutation End Up On Both Sides Of A Tumor? Left private mutations public Cells Mi
grate But Glands Donât Migrate Much ADENOMA ADENOMA CANCER CANCER Colorectal Tumors Benign Adenomas (born to be good?) Cancers: Invasive and Metastatic (born to be bad?) Mutation Patches Mutation Polka Dots 15 16 17 18 19 20 21 22 23 24 25 26 10 11 12 13 14 15
16 17 18 19 20 21 22 23 24 25 26 10 11 12 13 14 10 11 12 13 14 15 16 17 18 19 17 18 19 20 21 22 23 25 26 27 28 APC1338 Both 17 18 19 20 21 22 23 24 25 26 27 28 17 18 19 20 21 22 23 24 25 26 27 28 Adenoma Cancer public mutation private mutation private mutation
Microdissection Data Rate of new diagnoses and death in the Surveillance, Epidemiology, and End Results data from 1975 to 2005. Welch H G , and Black W C JNCI J Natl Cancer Inst 2010;102:605 - 613 Published by Oxford University Press 2010. Many Small Detecte
d âCancersâ Likely Will Not Kill Their Hosts Potential To Distinguish Early Lesions âBorn To Be Badâ From those âBorn To Be Goodâ Difficult To Predict The Lethality Of Small Human Tumors (lessons from screening) MODEL ASSUMPTIONS/TESTABILITY/PROBLEMS
/PROMISES 1) Start From Single Cell 2) Grows Into A Tumor (how fast?) 3) Presence or Absence of Early Mixing 1) Sample Single Glands 2) SNP microarrays 3) DNA sequencing 4) Specific Predictions 1) Implies A Burst Of Mutations During The First Few Divisions (Man
y Detectable Private Mutations) Assumptions Testability Problems Public Mutations (all tumor cells) 1) Implies The First Few Tumor Cells Divisions Are Unlike Any Other (Singularity) Promises Acknowledgements ⢠Yasushi Yatabe ⢠Kyoung - Mee Kim ⢠Jung Y
eon Kim ⢠Aimee Kang ⢠Peter Calabrese ⢠Kim Siegmund ⢠Paul Marjoram ⢠Simon Tavare ⢠Trevor Granham ⢠Christina Curtis ⢠Andrea Sottoriva current cell ( end ) zygote (start) Genomes Are âHistoricalâ Documents (almost perfect copies of c