
To the penny placed on your desk Observe the penny but dont touch it What happens Explain why 2 Give the penny a little push What happened Why did the penny move the second time and not the first time ID: 917275
Download Presentation The PPT/PDF document "Section 2.4 &2.5: Enzymes" is the property of its rightful owner. Permission is granted to download and print the materials on this web site for personal, non-commercial use only, and to display it on your personal computer provided you do not modify the materials and that you retain all copyright notices contained in the materials. By downloading content from our website, you accept the terms of this agreement.
Slide1
Section 2.4 &2.5: Enzymes
Slide2To the penny placed on your desk.
Observe the penny but don’t touch it. What happens?
Explain why
2. Give the penny a
little
push. What happened?
Why did the penny move the second time and not the first time?
What was added the second time and not the first time?
Slide3Energy is neither created or destroyed, it continues to be transferred.
All Chemical reactions involve a change/transfer in energy.
Chemical Reactions Release and Absorb Energy
Potential Energy
Potential to Kinetic Energy
Potential Energy
Potential Energy
Slide4Observing a Chemical Reaction
Put 50 mL of water into a flask
Add 10 drops of
Bromothymol
Blue
(
an acid indicator which will turn the solution
yellow if an acid is present)
Get the temperature in `C.Add one Alka
Seltzer tablet (split into two)Observe reaction and write down observations
Get the temperature after the reaction occurs.What you observed was:
CO
2 + H2O H2CO3 (Carbonic Acid) Reactants Product
Slide5Energy is needed to start a Chemical Reaction
Activation Energy
is the amount of energy that needs to be absorbed to start a chemical reaction
The push to get the boulder to go down the hill
Activation Energy
Slide6Do you have this bottle at home?
What is it?
What is it used for?
Can you keep it in your medicine cabinet forever?
Why?
Hydrogen Peroxide
Disinfecting a wound
NO!!
It will naturally break down into Water and Oxygen
Slide7What happened when the Manganese Dioxide was added to the 3% Hydrogen Peroxide?
Slide8What happened when the Manganese Dioxide was added to the 30% Hydrogen Peroxide?
Slide9How can we reduce the amount of activation energy needed to get a reaction started?
2H
2
O
2
2H
2
O + O2
This reaction will occur on its own but will take a long time and a lot of activation energy
Catalysts reduce the amount of activation energy necessary to get a reaction started
This is a method of cleaning contact lenses using a Manganese dioxide disk which will quickly break hydrogen peroxide into water in oxygen and get your contacts totally disinfected!!The Manganese dioxide disk can be used over and over again without a loss of functioning.
Slide10A Chemical Reaction with and without a catalystThe Catalyst
decreased the amount of activation energy needed to get the reaction started
a catalyst
a catalyst
Slide11Does anyone suffer from eating…?
…Beans, peanuts, broccoli, cauliflower, Brussel sprouts, cabbage, peppers, onions…
?
You know what I am talking about!!!
This happens because we can’t digest
alpha-galactose
present in these foods There is something to prevent this!! Beano contains the
Enzyme alpha-
galactosidase to allow us to break down alpha-galactose
Slide12What is the name of the sugar found in milk? Maybe you can’t tolerate lactose sugar and are lactose intolerant.
What do people do who are lactose intolerant?They take Lactaid pills which contain the enzyme
Lactase
We will do a lab on this
Enzymes
are
protein catalysts
made by living organisms that
reduce the amount of activation energy necessary to start and control the rate of a chemical reaction
Lactose
Slide13Our cells always produce Hydrogen Peroxide. 2H
2
O
2
2H
2
O + O2
This is highly toxic to our cells.An enzyme called catalase
will allow hydrogen peroxide to quickly break down to water and oxygen. You have seen this with a cut and blood when you put hydrogen peroxide on it.
One catalase molecule can convert 40 million of molecules of hydrogen peroxide to water and oxygen each second
Slide14Characteristics of Enzymes
Made up of
Proteins
Are specific
to whatever they act upon
Are
reusable
– don’t get used up in a chemical reactionUsually have names ending in “ase
”Enter directly into a reaction
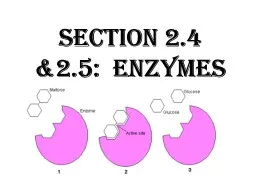
Slide15How Enzymes workTheir shape is specific to what they act upon (the
substrate
)
They will bind directly to the
substrate
as an
enzyme-substrate complex
The binding site is called the
Active SiteA substrate and its enzyme fit together like puzzle pieces –
Lock and Key Theory
Slide16Slide17Substrate
Slide18What can influence an enzymes reactivity?
Temperature
Since enzymes are proteins, their structure can be changed or destroyed by high temperatures.
As temperature increases, the rate of enzyme activity increases to a point then quickly decreases.
Study Island Virtual lab
In us, this temperature is at ~ 40`C
Slide19pH Too high or too low pHs will effect the enzyme activity and possibly change its shape.
Lactase activity virtual lab
Different for different organs
Slide20Concentration of enzymes As the number of enzymes increases, the rate of reaction increases also to a point
where all the enzymes are already bound to an active site of a substrate.Additional enzymes won’t speed up a reaction at this point.
Slide21ReviewTo which biomolecule group do enzymes belong to and usually end in …?
2. What is an enzyme?
What do enzymes do?
4. What part of the enzyme enters into a reaction?
Proteins, -
ine
Organic catalyst
Controls the rate of a reaction
Active Site
Slide22What is the term used for the molecule which is acted on by the enzyme?When the two are together, what is it now called?
What will extreme pH’s and temperatures do to the active sites of enzymes?
What is the ideal pH and temperature for most reactions in our body?
Enzyme-Substrate Complex
Denature the active site – Change the shape
~6.5 – 7.4 & 37`C
Substrate
Slide23Label the diagram using the terms, Product, substrate, enzyme, active site and, enzyme-substrate complex.