PPT-Certification Program Requirements*
SO
stefany-barnette
Published 2018-11-15 | 4974 Views
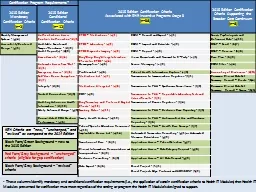
2015 Edition Certification Criteria Associated with EHR Incentive Programs Stage 3 n38 2015 Edition Certification Criteria Supporting the Broader Care Continuum
Download Presentation
Download Presentation The PPT/PDF document "Certification Program Requirements*" is the property of its rightful owner. Permission is granted to download and print the materials on this website for personal, non-commercial use only, and to display it on your personal computer provided you do not modify the materials and that you retain all copyright notices contained in the materials. By downloading content from our website, you accept the terms of this agreement.
