PPT-Kinetic Time Scales AU-MSP Module
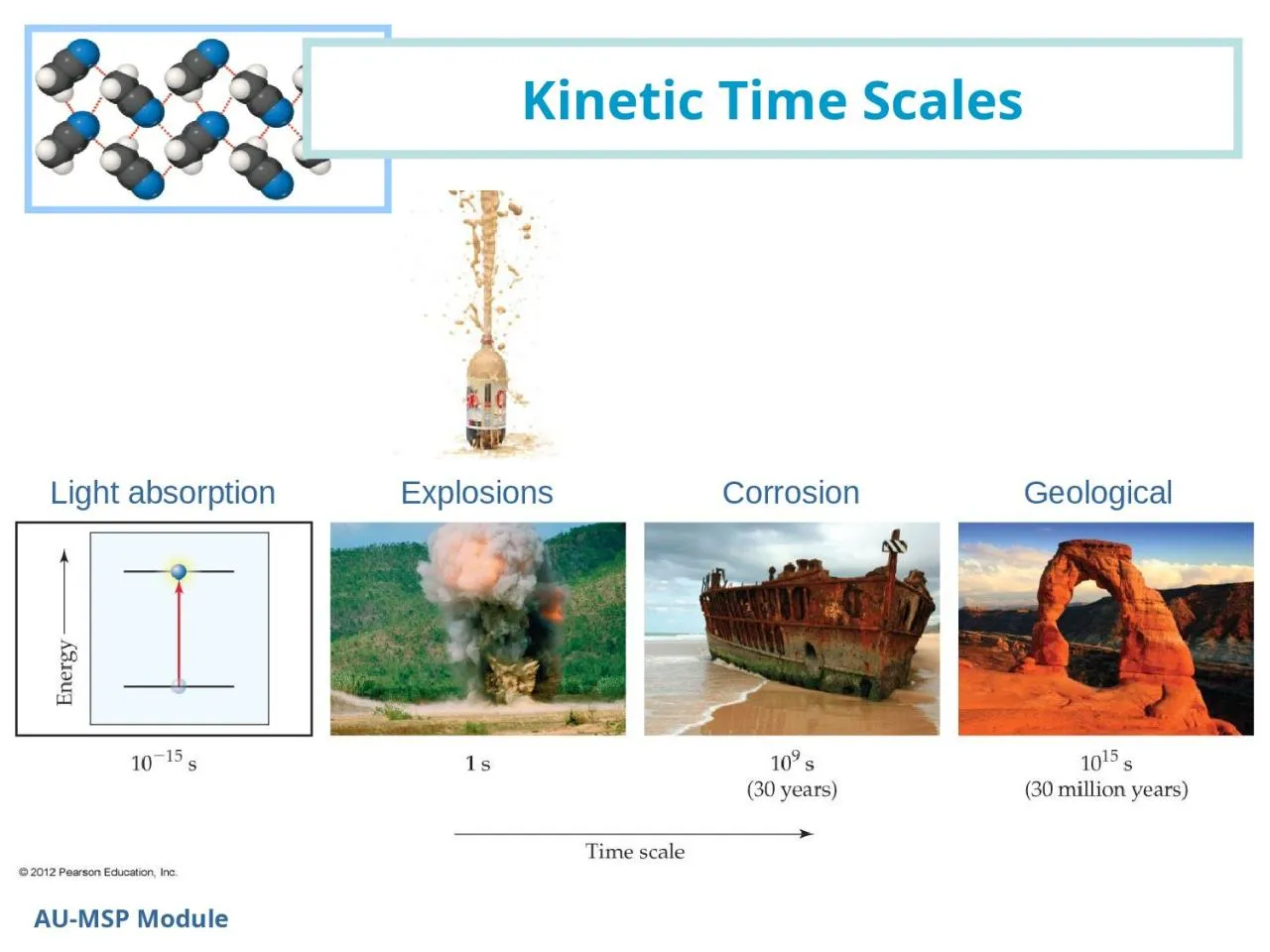
Light absorption Explosions Corrosion Geological Energy Barrier in Reactions AUMSP Module Chemical and physical reactions have energy barriers or an activation energy
Download Presentation
"Kinetic Time Scales AU-MSP Module" is the property of its rightful owner. Permission is granted to download and print materials on this website for personal, non-commercial use only, provided you retain all copyright notices. By downloading content from our website, you accept the terms of this agreement.
Presentation Transcript
Transcript not available.