PPT-SOF/VEL/VOX
SO
pamella-moone
Published 2018-01-15 | 5274 Views
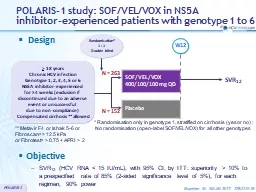
400100100 mg QD N 263 N 152 W12 Placebo gt 18 years Chronic HCV infection Genotype 1 2 3 4 5 or 6 NS5A inhibitorexperienced for 4 weeks exclusion if discontinued
Download Presentation
Download Presentation The PPT/PDF document "SOF/VEL/VOX" is the property of its rightful owner. Permission is granted to download and print the materials on this website for personal, non-commercial use only, and to display it on your personal computer provided you do not modify the materials and that you retain all copyright notices contained in the materials. By downloading content from our website, you accept the terms of this agreement.
