PPT-Statin Side effects  Active treatment
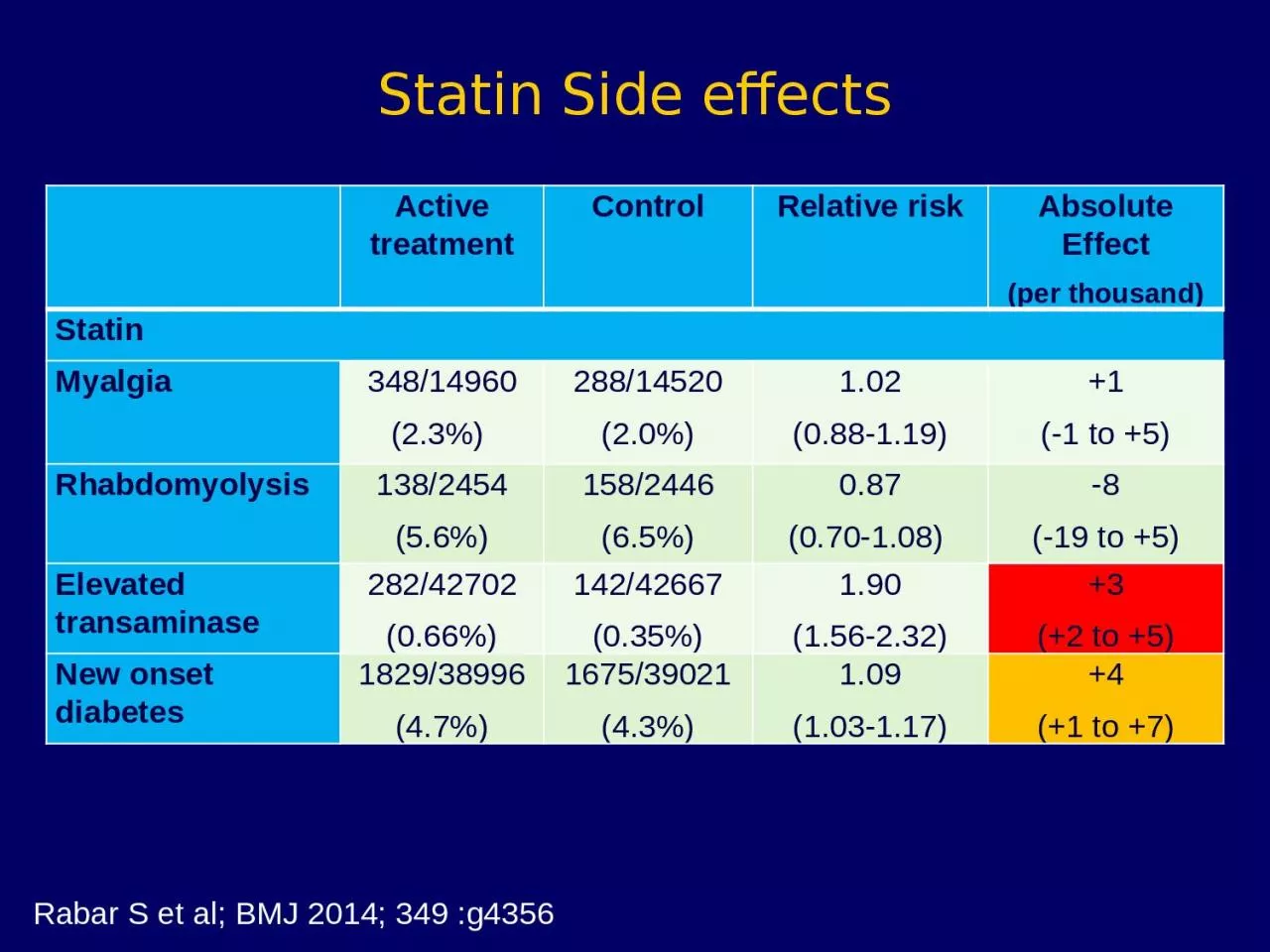
Control Relative risk Absolute Effect per thousand Statin Myalgia 34814960 23Â 28814520 20 102 088119 1 1 to 5 Rhabdomyolysis 1382454 56 1582446 65
Download Presentation
"Statin Side effects  Active treatment" is the property of its rightful owner. Permission is granted to download and print materials on this website for personal, non-commercial use only, provided you retain all copyright notices. By downloading content from our website, you accept the terms of this agreement.
Presentation Transcript
Transcript not available.