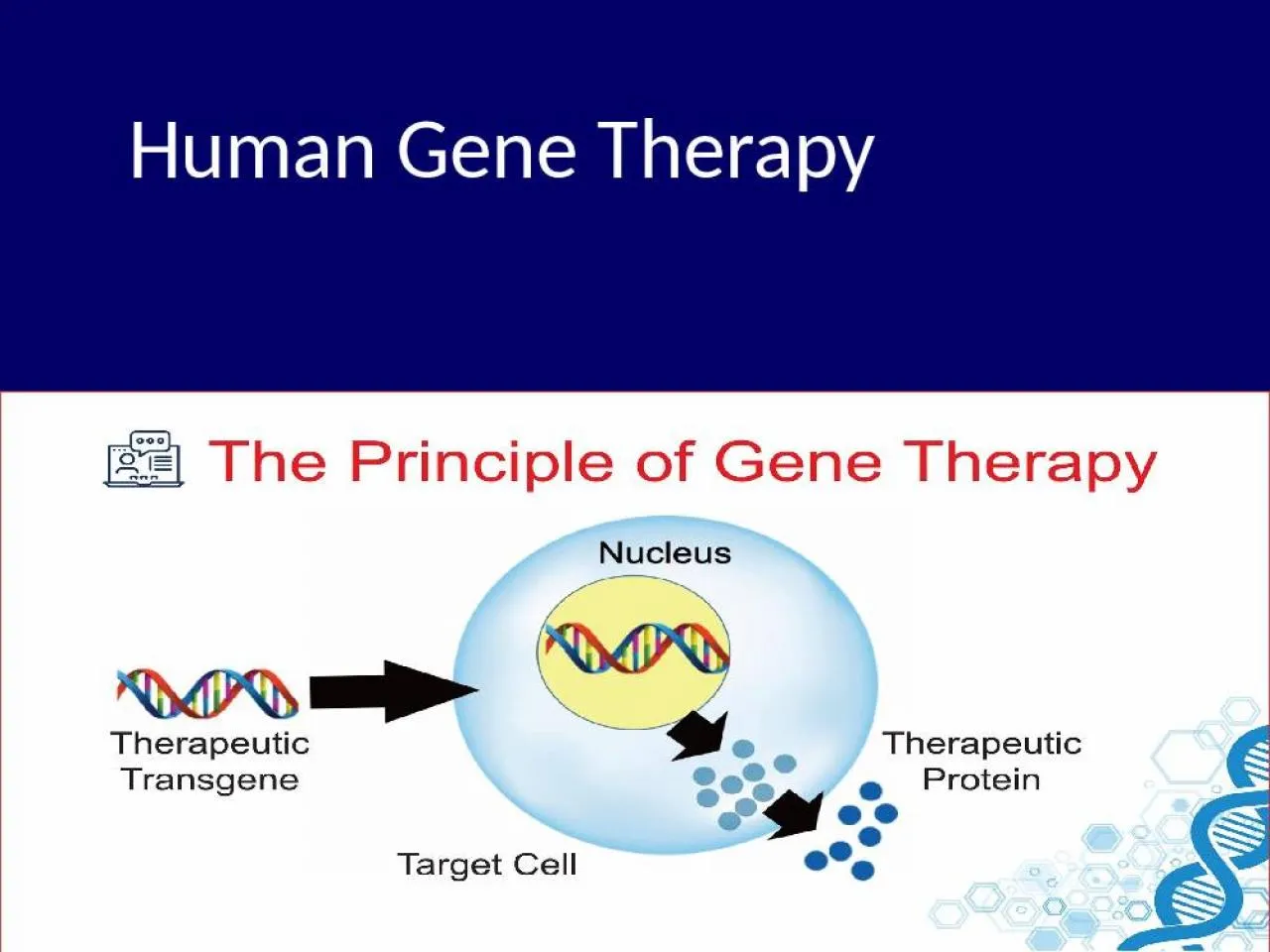
PPT-Human Gene Therapy Introduction
Genetic Counseling Treating Genetic Disease Removing an affected body part Replacing an affected body part or biochemical with material from a donor Delivering pure
Download Presentation
"Human Gene Therapy Introduction" is the property of its rightful owner. Permission is granted to download and print materials on this website for personal, non-commercial use only, provided you retain all copyright notices. By downloading content from our website, you accept the terms of this agreement.
Presentation Transcript
Transcript not available.