PPT-GENETIC DISORDERS DISEASES
SO
natalie
Published 2022-06-20 | 4904 Views
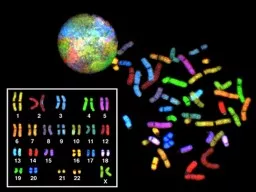
GENETIC ENVIRONMENTAL BOTH CONGENITAL HEREDITARY FAMILAL MUTATIONS PERMANENT change in DNA GENE MUTATION may and often result in a single base error CHROMOSOME MUTATION
Download Presentation
Download Presentation The PPT/PDF document "GENETIC DISORDERS DISEASES" is the property of its rightful owner. Permission is granted to download and print the materials on this website for personal, non-commercial use only, and to display it on your personal computer provided you do not modify the materials and that you retain all copyright notices contained in the materials. By downloading content from our website, you accept the terms of this agreement.
