Search Results for 'Report Of Adverse Events Following'
Report Of Adverse Events Following published presentations and documents on DocSlides.
REPORT REPORT REALISING AMBITION TITLE OF REPORT STATIC OR NOT TBC YEAR REPORT CONTENTS TITLE OF REPORT STATIC OR NOT TBC TITLE OF REPORT STATIC OR NOT
57347E57347LQFUHDVLQJ57347LQYHVWPHQW57347LQ57347WK...
Progess report Progress report Progess report An update on WHOs work on female genital mutilation FGM Progress report An update on WHOs work on female genital mutilation FGM Introduction Female genita
In the world today there are an estimated 130140 ...
Container Loading Supervision Report-Loading Survey Inspection Report
Container Loading Supervision Report-Loading Surv...
Report Writing Unit III What is a business report?
A business report is an oral presentation or writ...
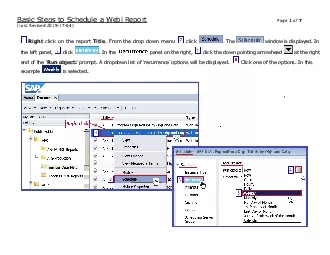
a Webi Report
Basic Steps to Schedule Page 1of 7Last Revised 201...
A Report on Mental Illnesses in CanadaA Report on Mental Illnesses in
31313131 CCCHAPTER HAPTER HAPTER 222MMMMOOD OOD OO...
Writing of Management Audit Report Report Writing
Report Writing. . Dinesh Pant. Process of Managem...
The inheritance of Abraham A report on May Introducti on Ten years ago the General Assembly received the report Theology of Land and Covenant from the Board of World Mission Church and Nation Commi
This report concluded with 5763057718576165772557...
Report on App Platform and Device Preferences from the Leader in Secure Mobility GOOD TECHNOLOGY TM MOBILITY INDEX REPORT Q This report is part of the Good Technology Mobility Index an ongoing initia
This document presents the details of the Good Mo...
Neustar Annual DDoS Attacks and Impact Report THE DANGER DEEPENS The Danger Deepens Neustars Annual DDoS Attacks and Impact Report Welcome to the DDoS Attacks and Impact Report For the third conse
What were their experiences in 2013 The results s...
Spring Report Dice or No Dice The Casino Debate in Massachusetts University of Massachusetts Boston College of Management Financial Services Forum Spring Report Spring Report Dice or No Dice The
The Financial Services Forum is a source of curre...
Credit Report Basics Your credit report and previous searches This fact sheet explains about previous searches on your credit report
One year on Experian and Equifax two years on Cal...
Life Insurer Financial Report Includes Custom Sort Report Sorted By Com dex Ranking Rating Scales Report Stan The A nnuity Man P
O Box 2225 Ponte Vedra Beach FL 32004 Phone 904614...
Life Insurer Financial Report Includes Custom Sort Report Sorted By Com dex Ranking Rating Scales Report Stan The A nnuity Man P
O Box 2225 Ponte Vedra Beach FL 32004 Phone 904614...
Table of Contents Page Fiji Population Census Fiji Isla nd Bureau of Statistics FIBoS HEALTH IN FIJI Ministry of National Planning MDG Report nd Report Report for the Fiji Islands Health I
Vol1 No1 2010 unpublished 2007 Fiji Situation Rep...
YOUR EQUIFAX CREDIT REPORT YOUR EQUIFAX CREDIT REPORT Data Protection Act YOUR EQUIFAX CREDIT REPORT Credit reference agencies Credit reference agencies hold public information such as electoral ro
The credit reference agencies dont hold blacklist...
CREDIT BUREAU INFORMATION BECU To Report Fraud To dispute information reporte d by BECU on credit report Equifax www
equifaxcom To Report Fraud To Update Fraud Informa...
Life Insurer Financial Report Includes Custom Sort Report Sorted By Com dex Ranking Rating Scales Report Stan The A nnuity Man P
O Box 2225 Ponte Vedra Beach FL 32004 Phone 904614...
Application Report SLAA November Interfacing the MSP and TLC AD Converters Mark Buccini Mixed Signal Controllers ABSTRACT This application report describes how to interface an MSP mixedsignal micro
This report is written for the MSP430x11x1 family...
An HCPro Publication Coder Productivity Benchmarks A Special Report Coder Productivity Benchmarks A Special Report Coder Productivity Benchmarks A Special Report Dear reader Establishing coder produ
Do your current fulltime equivalent FTE employees...
Shaping the Future of South Australia A summary of the Top identified actions August Report Disclaimer Inherent Limitations This report has been prepared by KPMG at the request of the Committee f
This report has been prepared for general guidanc...
ORMAT OF THE REPORT FOR THE FINAL YEAR PROJECT Page of Guideline for Final Year BE th Seme ster Project Report Submi ssion or ll the final year students who are working on their th semester project
The Report is to be submitted whether the project...
News consumption in the UK Report Research Document Publication date June News Consumption in the UK Report About this document This report provides key findings from Ofcoms research into news c
It also examines changes to how UK adults have co...
CDFA Grape Crush Report Final Crop GRAPE CRUSH REPORT OVERVIEW Information contained in this Report was supplied by processors to fulfill the reporting requirements of Section
5 of the Food and Agricultural Code The Final Grap...
Commonwealth of Massachusetts Motor ehicle Crash Operator Report When Should You File a Report You should file a report if youre the operator of a vehicle involved in a crash where the damage to any
You should file the report within 5 days of the d...
Special Report Special Report Center for Applied Resea
C Fall 2006 his report summarizes findings from a ...
Reviewing Another Institution’s Appraisal Report
Presenter. Craig Benton, MAI. Director, Valuation...
Dream Report®
Programming Free™. Designer Friendly™ . REPOR...
More Than a Report
Bringing your reports to life. !. Presented by Br...
The Essentials of Bedside Shift Report
in Acute Care. Adapted from AHRQ and the . Stu...
ROSI Express Report Training:
Scheduled Courses with Instructor/Coordinator Dia...
Annualreport
report 2015 strategicreport report financial2015 f...
The South Asian Report on the Child-friendliness of Governm
South Asian Report . On the Child-friendliness of...
Report from the
CLIC-ILC General Issues Working Group. Mike Harri...
Report writing
RB, pp 17-26. What is a report?. . A written sta...
Lab report resubmission
Did not meet proficiency: under 70%. Must resubmi...
Elementary Report Card
Revisions. Franklin Public Schools. Parent Inform...
Bedside Report Plan
Research Poster Presentation. Jordan Furstenau. F...
To Report or Not to Report?
What do you think of when you see these words?. S...
Technical Report Documentation Page1. Report No. FHWA-CFL/TD-06-0
5. Report Date November 2006 4. Title and Sub...